Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Lesions (cancerous)
Author: Vanessa Ngan, Staff Writer, 2003.
Introduction
How it works
Uses
Prescription
Precautions
Side effects
Storage and handling
Metvix movie
MAL-PDT using natural daylight
Methyl aminolevulinate (MAL) is a topical photosensitising agent that is used in photodynamic therapy (PDT). It is available as a 16% strength cream in a 2g tube. The trade name is Metvix™.
When applied to the skin, methyl aminolevulinate is selectively absorbed into cancer cells. The drug is converted into photoactive porphyrins, naturally-occurring intracellular photosensitising chemicals that bind with iron to make haemoglobin. When cancer cells full of porphyrins are exposed to specific wavelength of light (570–670 nm), a molecular reaction occurs that results in destruction of the cancer cells.
Methyl aminolevulinate is classed as an antineoplastic agent (anti-cancer drug) and is approved for use in New Zealand for the treatment of:
MAL-PDT is recommended for lesions that are unsuitable for other available therapies due to possible complications and/or poor cosmetic outcome. It may be particularly useful for lesions on the mid-face or ears, lesions on severely sun damaged skin, large lesions or recurrent lesions.
Clinical trials around the world using MAL-PDT have shown it to have comparable cure rates to other conventional treatments such as cryotherapy or surgery. A distinct advantage of MAL-PDT is that it selectively targets tumour cells. This means that surrounding healthy cells are not affected by the treatment thus leaving minimal scarring and a much better cosmetic outcome.
MAl-PDT involves a two-stage process. The first stage is application of the methyl aminolevulinate cream.
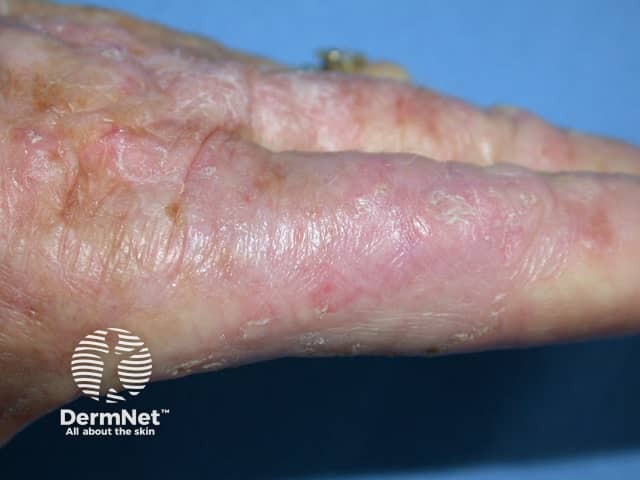
Actinic keratoses
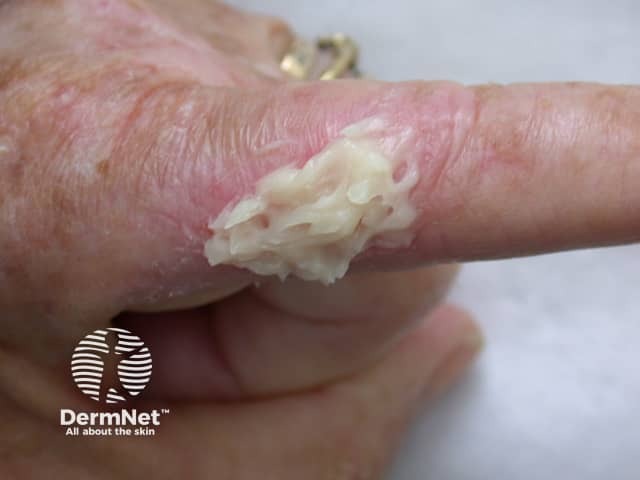
Metvix cream
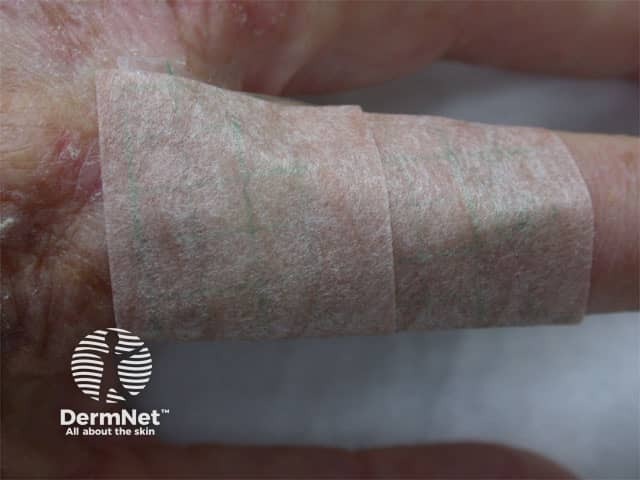
Taped treatment area


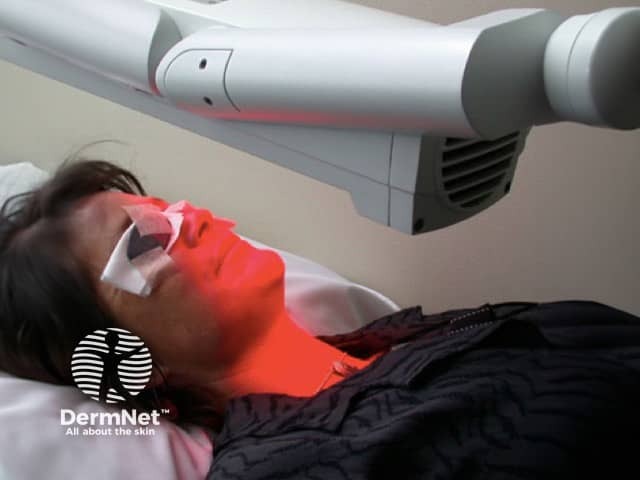
The second stage involves exposing the lesion to light.
After treatment the lesions will be covered with an occlusive dressing for at least 24 hours. Lesions usually heal completely within one to four weeks. More than one lesion can be treated in a single treatment session. A repeat second treatment may be done approximately one week later: this is recommended for most basal cell carcinomas and thicker actinic keratoses.
The practitioner should check the lesions after about 3 months. If they show a non-complete response, re-treatment may be necessary.
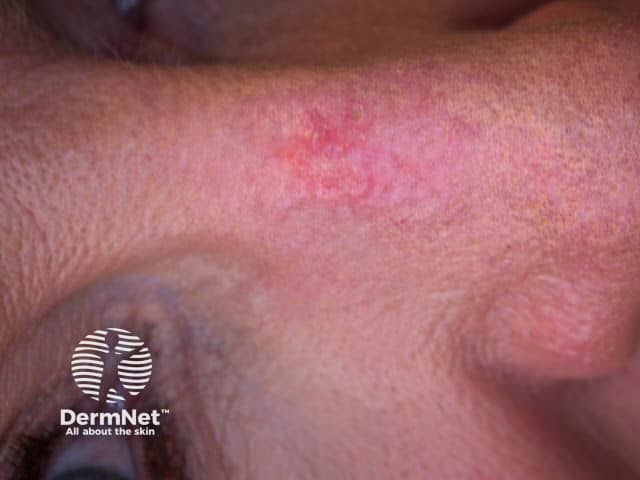
Prior to treatment
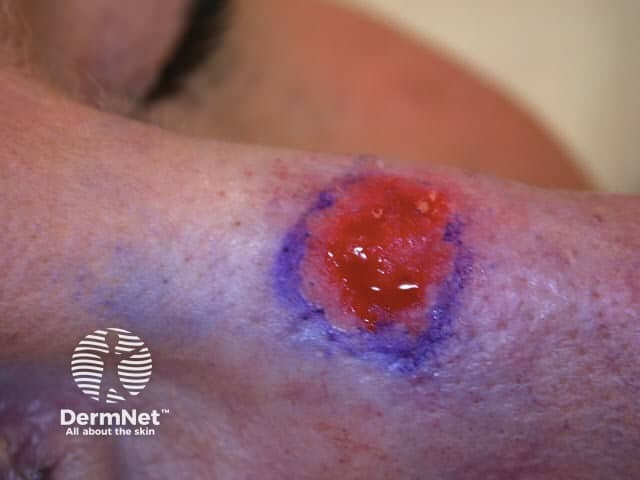
Lesion scraped
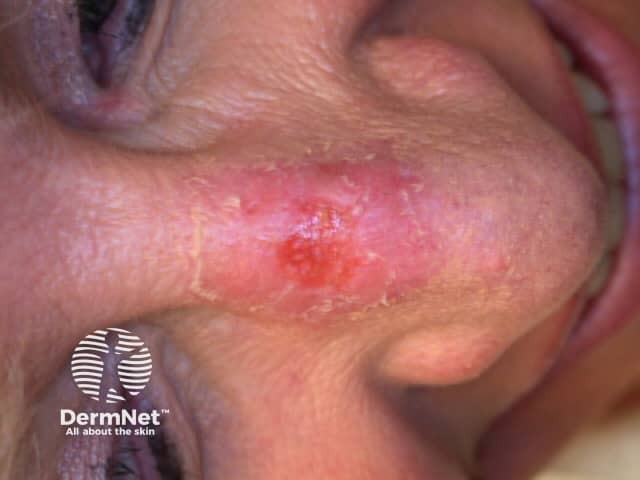
One week after MAL PDT
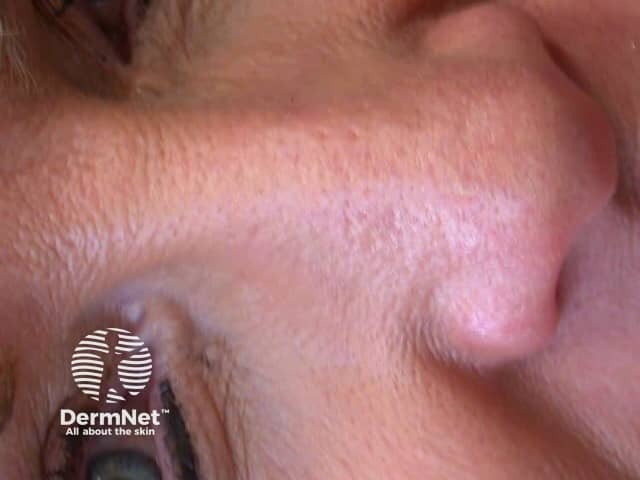
One month after MAL PDT
Pain threshold varies between individuals. It is mediated by induction of free radicals by protoporphyin IX.
Topical anaesthesia is not effective. Local (dermal) infiltration of anaesthetic should be avoided due to theoretical reduction if efficacy of treatment due to reduced oxygenation of tissue. Subcutaneous anaesthesia and nerve blocks in expert hands may be beneficial.
Approximate 60–80% of patients undergoing MAL-PDT experience some side effects. These are usually related to the treated area being sensitive to light and are like sunburn. Side effects are of mild to moderate intensity and usually subside within a few days after treatment.
As the skin cancers cells die off, the lesion may blister and ulcerate. This generally heals within 4 weeks.
Very common
Common
Uncommon
Daylight-mediated PDT is a convenient form of treatment of thin actinic keratoses on the face and scalp. Results have been shown to be as good as with conventional MAL-PDT (described above). Treatment can be undertaken during spring, summer or autumn in most parts of the world.
The natural daylight PDT procedure is as follows:
Natural daylight PDT is followed by sunburn-like inflammation in treated areas for 5 to 8 days. The skin is red, and may become blistered and crusted. The reaction tends to be most severe at 3 to 4 days after treatment. Redness can be reduced by pre-treatment with topical steroid.
Similar cure rates as conventional MAL-PDT and excellent cosmetic results are described for thin actinic keratoses. Advantages of natural daylight PDT include:
Daylight MAL-PDT may also be undertaken indoors. Lux should be > 5,000 for it to be effective in the treatment of actinic keratoses. Options include:
New Zealand approved datasheets are the official source of information for these prescription medicines, including approved uses and risk information. Check the individual New Zealand datasheet on the Medsafe website.
Medical Practitioners may contact the sponsor Galderma for further information about Metvix™ Cream PDT.