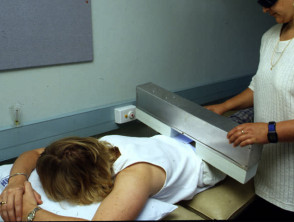
What is UVB phototherapy?
Ultraviolet B (UVB) phototherapy delivers shortwave ultraviolet radiation to treat skin conditions such as psoriasis. Phototherapy works by suppressing DNA synthesis, which in turn reduces inflammation.
Narrowband UVB phototherapy (311–312 nm) is increasingly used compared to broadband UVB phototherapy (270–350 nm). This is because exposure and treatment times are shorter and result in longer periods of remission.
How is UVB phototherapy delivered?
UVB phototherapy is delivered within a specially designed cabinet. Fluorescent light tubes are positioned to target affected areas with ultraviolet radiation (eg, hands or feet). For full body treatments, patients are undressed with genitalia covered, and eyes or full face should be protected with goggles or a face mask.
Most patients will have treatment three times a week, at least 24 hours apart. Session length is initially ≤5 minutes, and may be uptitrated as required to a maximum of 30 minutes per session. The number and length of sessions vary for each individual, taking into account their skin type, skin condition, and previous response to treatment.
Phototesting to determine the minimum dose that produces skin redness (MED testing) is recommended and a starting dose of 70% of the MED is often used. The dose is escalated by 20% each treatment, provided there are no side effects or episodes of burning.
Targeted phototherapy devices (eg, excimer laser, brush or comb devices) have also been developed as treatment options for more challenging areas while sparing uninvolved skin.
What is UVB phototherapy used for?
Common indications:
- Psoriasis
- Dermatitis eg, atopic dermatitis (eczema)
- Vitiligo.
Other indications:
- Nodular prurigo
- Cutaneous T-cell lymphoma
- Pityriasis lichenoides
- Symptomatic dermographism
- Lichen planus
- Mycosis fungoides
- Polymorphic light eruption
- Cutaneous graft versus host disease
- Mastocytosis
- Generalised pruritus.
Psoriasis
Psoriasis is a chronic inflammatory skin disorder, which varies considerably in extent and severity. UVB is suitable for most people with extensive psoriasis. It may not suit those with very fair skin or those whose psoriasis gets worse in sunlight.
Most patients will have clearance or improvement in their psoriasis after 12 to 24 treatments. After treatment is discontinued the skin may remain clear for months. If psoriasis flares up again, further UVB treatment may be necessary.
Dermatitis
UVB is occasionally used for severe cases of dermatitis, especially atopic dermatitis (eczema). Frequency and dosage of treatment are usually longer than for psoriasis.
Vitiligo
UVB is effective in treating vitiligo by immune suppression and stimulation of cytokines. Treatment is given twice a week for 3–4 months and continued until re-pigmentation is complete or for 1–2 years. It may take several months to see an improvement and caution must be taken with fair skin.
What are the contraindications to UVB phototherapy?
Absolute contraindications:
- Previous melanoma or skin cancer
- Familial atypical multiple mole and melanoma (FAMMM) syndrome (also known as dysplastic naevus syndrome); see Atypical melanocytic naevus
- Systemic lupus erythematosus
- Dermatomyositis (juvenile or adult-onset)
- Genetic conditions that predispose an individual to skin cancer (eg, xeroderma pigmentosum, Gorlin syndrome/basal cell naevus syndrome)
- Bloom syndrome or Cockayne syndrome
- Patients unwilling or unable to comply with safety procedures; or who are medically unfit and unable to stand (eg, severe cardiovascular or respiratory disease).
Relative contraindications:
- History of exposure to arsenic/ionising radiation
- Pregnancy (if used folic acid supplements should be taken and serum levels measured)
- Current pre-malignant skin condition
- Concomitant immunosuppressive therapy (ciclosporin, azathioprine, mycophenolate)
- Photo-induced epilepsy
- Cataracts
- Bullous pemphigoid/pemphigus
- Significant liver dysfunction
- Age <16 years.
What are the benefits of UVB phototherapy?
- Can be used to treat the whole body or localised areas.
- Narrowband UVB phototherapy is less likely to cause cancer than PUVA (photochemotherapy) as there is less UV exposure and a narrower range of UV radiation.
- In some places, home phototherapy may be offered.
- Safer and often cheaper alternative to systemic therapy.
What are the disadvantages of UVB phototherapy?
- Requirement to attend regular appointments three times a week, unless home phototherapy is available.
What are the side effects and risks of UVB phototherapy?
Short-term side effects:
- Dry skin
- Itchy red rash 6–24 hours after treatment
- Sunburn may sometimes occur
- Reactivation of herpes simplex virus – cold sores may be prevented by sun protective lip balm
- Worsening of skin disease
- Provocation of photodermatoses
- Increased pigmentation to treated area/s.
Long-term side effects:
- Skin ageing
- Increased risk of skin cancer; those who have received more than 500 treatments may be considered for skin cancer surveillance
Instructions for patients receiving UVB phototherapy
- Keep all scheduled appointments. Allow time for changing.
- Tell phototherapy clinic staff about any medical conditions (including eye disease), and if you take any medications (as some may make you more prone to burning).
- Do not apply any ointments or cosmetics (including perfumes and coal tar) except as directed by your doctor or phototherapist.
- Follow your phototherapist’s advice re. application of moisturisers or sunscreens. Some products may prevent absorption of UVB if applied before treatment. In general, they should be avoided for 2 hours before the phototherapy exposure, but should be applied after the treatment.
- Avoid other sources of exposure to ultraviolet light including natural sunlight and tanning beds.
- During treatments, eye or full face UV protection with goggles or a face mask should be used, and genitalia covered
- Notify the phototherapy team if you experience any side effects.
- Ensure you are attending regular appointments with your dermatologist.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).