Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Last Reviewed: August, 2025
Authors: Dr Mehak Chadha, Dermatology Registrar; Dr Laura Savage, Consultant Dermatologist, Leeds Centre for Dermatology, West Yorkshire, United Kingdom (2025)
Peer reviewed by: Nancy Huang (MBChB), DermNet Medical Writer, New Zealand (2025)
Previous contributors: Vanessa Ngan, Staff Writer; Dr Amanda Oakley, Dermatologist, New Zealand (1997); Dr Abdulhadi Jfri, Dermatology Resident; Dr Elizabeth O’Brien, Assistant Professor of Dermatology, McGill University, Montreal, Canada (2015)
Reviewing dermatologist: Dr Ian Coulson
Edited by the DermNet content department.
Introduction
Demographics
Causes
Clinical features
Severity assessment
Complications
Diagnosis
Differential diagnoses
Treatment
Outcome
Hidradenitis suppurativa (HS) is a chronic inflammatory skin disorder that affects apocrine gland-bearing skin, typically the axillae, groin, perianal region, buttocks, and inframammary folds. It is characterised by persistent, deep-seated, painful nodules and abscesses that can progress to sinus tract formation, purulent discharge, and scarring.
Also known as acne inversa, this disorder can significantly impact quality of life and is associated with several disease-modifying comorbidities, including metabolic syndrome and psychiatric disorders.
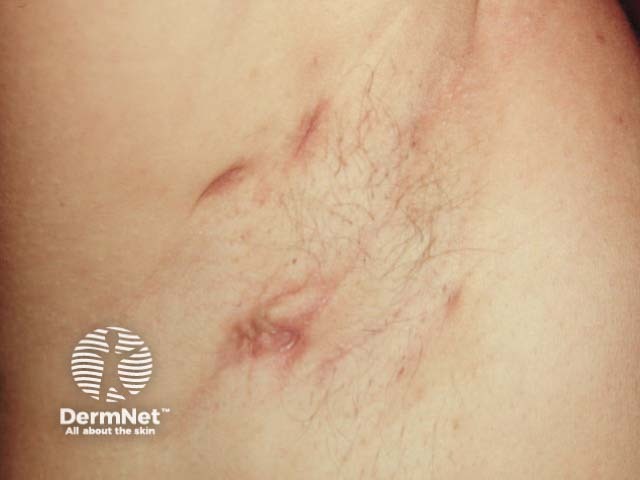
Mild axillary HS showing scars and post-inflammatory erythema
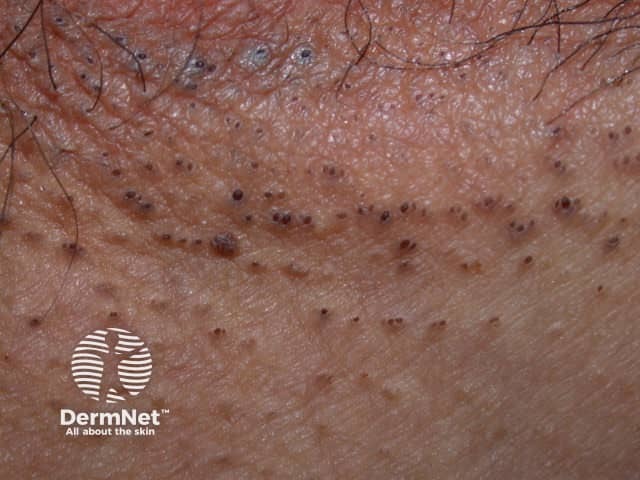
Marked comedones in axillary HS
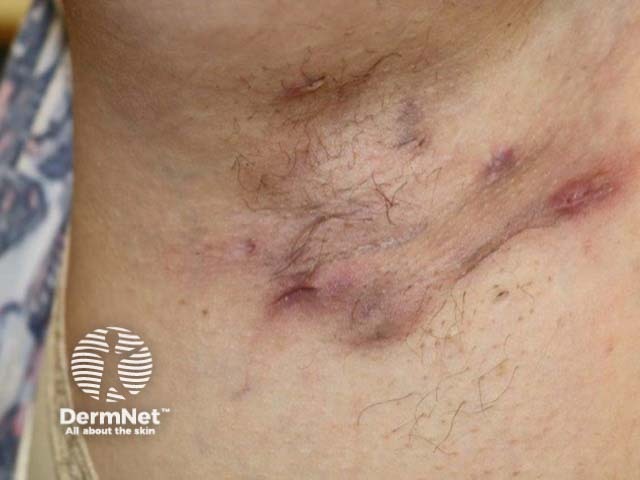
Nodules and sinus formation in axillary HS
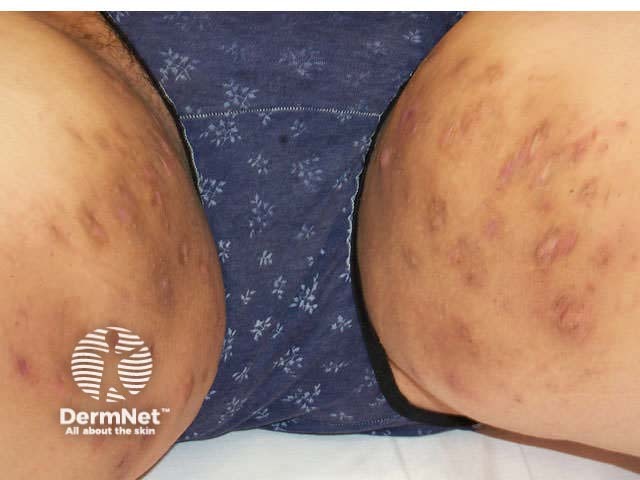
HS in the groins spreading down to the upper thighs
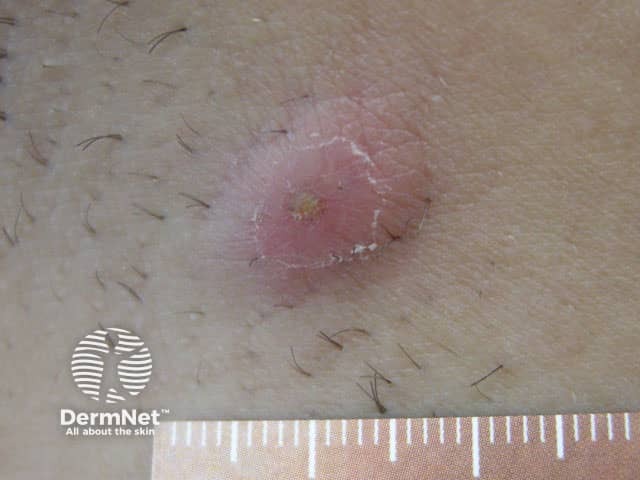
An acute painful inflammatory nodule in axillary HS
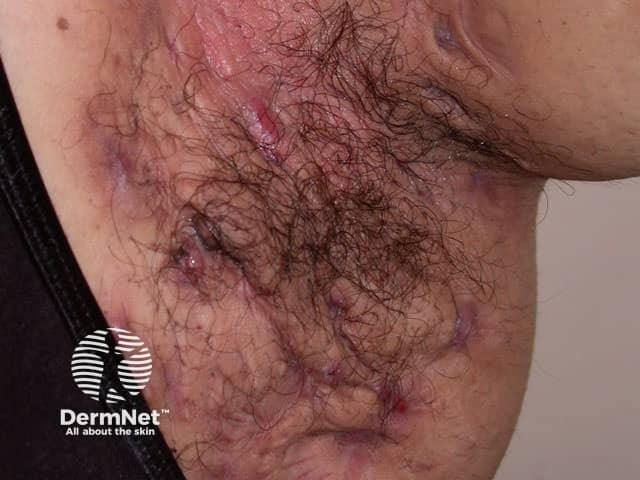
Severe axillary HS with scars and sinus formation
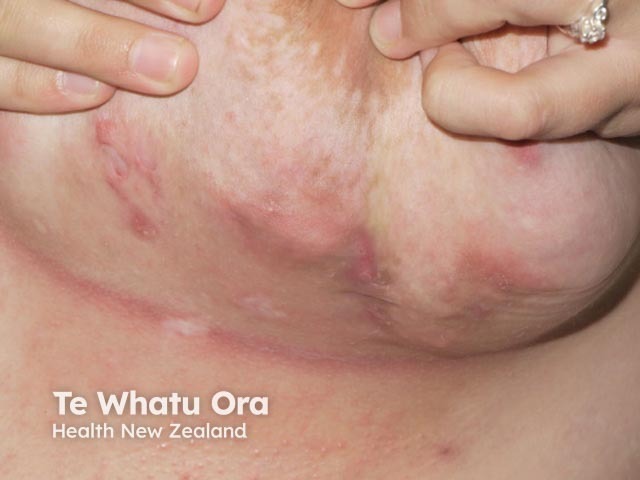
Submammary HS
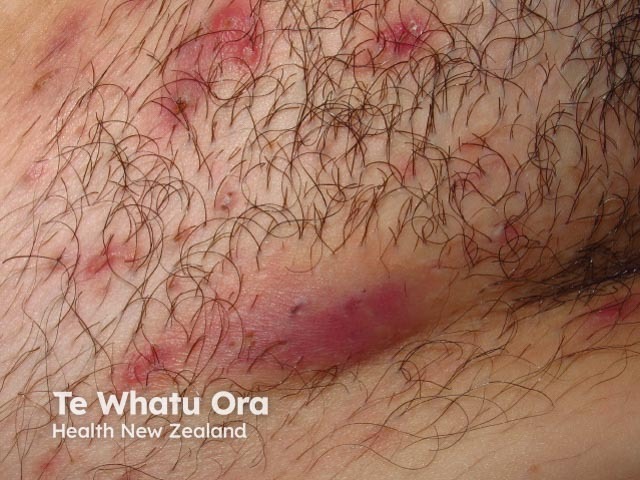
Inflammatory HS in the groin with multiple painful nodules
The global prevalence of hidradenitis suppurativa is estimated to range from 0.1% to 4%. However, underdiagnosis and misdiagnosis likely mean that the actual prevalence is higher. Delayed diagnosis is also common.
HS typically presents following puberty by the second to third decades of life. Studies show that there is no racial predilection, although some surveys suggest it may be more common in African Americans.
Risk factors for the development of HS:
Associated comorbidities:
The exact cause of hidradenitis suppurativa is not fully understood. HS is described as an autoinflammatory syndrome, and its development is influenced by genetic, immune, hormonal, microbial, mechanical, and lifestyle factors. Hidradenitis suppurativa is not contagious, and it is not caused by poor hygiene.
The early pathogenesis of HS is thought to involve follicular occlusion and rupture, an abnormal innate immune response, and a subsequent inflammatory cascade.
Leukocytic infiltration, oedema, and bacterial dysbiosis within ruptured follicular units lead to inflammatory nodules and abscesses.
Persistent inflammation results in extensive fibrosis, and late signs of HS consist of epithelial-lined, pus-draining tunnels deep in the dermis.
Hidradenitis suppurativa is characterised by:
Hidradenitis suppurativa typically affects intertriginous areas and can affect one or multiple sites:
Hidradenitis suppurativa can also occur within rare autoinflammatory syndromes:
There are several severity scales to assess hidradenitis suppurativa clinically and academically.
This is the most commonly recognised clinical scoring system:
The Hurley staging system is not a dynamic means of assessment. Once a stage is reached, it cannot be downgraded due to the irreversibility of scarring with medical therapy.
IHS4 is a dynamic scoring tool that can be particularly helpful in monitoring disease progression and response to treatment.
Number of nodules |
x1 |
Number of abscesses |
x2 |
Number of draining sinus tracts (fistulae/sinuses) |
x4 |
The final score is calculated by the weighted sum of number of nodules, abscesses, and draining sinus tracts.
Mild: <3 points
Moderate: 4-10 points
Severe: >11 points
This score is made by counting involved regions, nodules, and sinus tracts. Its use has declined due to its complexity and the introduction of other measures such as the IHS4.
Anatomical regions involved: axilla, pectoral, inguinal/genital, intergluteal, buttocks |
3 points for each region |
Types of lesions |
4 points for each abscess or draining fistula |
Distance between lesions OR size of single lesion |
8 points if >10 cm |
Are all lesions clearly separated by normal skin? |
6 points for no |
The HiSQOL is used to consider the degree of pain, the number of flares, and the impact on daily life in hidradenitis suppurativa. This is a reliable and valid tool to measure HS-specific health-related quality of life.
HiSCR is defined as a reduction in inflammatory lesion count (ie, abscesses, inflammatory nodules) and no increase in abscesses or draining fistulas, when compared with baseline.
It is predominantly used in clinical trials as a dichotomous outcome to assess the effectiveness of treatment with biologics. It has limited use in clinical practice.
The most commonly used scores are: HiSCR50, HiSCR75, and HiSCR90, representing a 50%, 75%, and 90% reduction in inflammatory lesion counts as compared to baseline, respectively.
Hidradenitis suppurativa is primarily diagnosed clinically, based on the modified Dessau criteria.
Diagnosis requires fulfilment of all three components:
Diagnosis is supported by:
Biopsy is not typically needed, but it can help exclude other conditions eg, if there are features suggestive of squamous cell carcinoma. Histological features of HS include:
Imaging is not necessary for diagnosis. However, ultrasound can help identify subclinical features, and MRI is useful for assessing complications and the extent of anogenital disease.
Optimal management of hidradenitis suppurativa requires a multidisciplinary team approach consisting of dermatology, general and/or plastic surgery, general practice, wound care management, dietetics, weight management services, and psychology.
Treatment should be tailored to each individual and follow local or regional management guidelines, where available.
Outlined below are commonly utilised treatments for hidradenitis suppurativa.
May be used alone for mild HS or as adjunctive therapies in more severe disease:
Triamcinolone acetonide 10–40 mg/mL injections to individual inflammatory lesions; may require repeat sessions on a weekly to three-monthly basis.
Antibiotic |
Drug and dosage |
Indications |
||||||
|---|---|---|---|---|---|---|---|---|
Oral tetracyclines |
Options:
|
Used for mild to moderate disease (Hurley Stage I/II) |
||||||
Clindamycin plus rifampicin |
Oral clindamycin 300 mg BD and rifampicin 300 mg BD for up to 12 weeks.
|
Used for severe disease (Hurley Stage III) |
||||||
Intravenous antibiotics |
IV ertapenem 1 g OD for 6 weeks. |
Used as rescue therapy for severe flares unresponsive to clindamycin plus rifampicin. |
||||||
Biologics should be initiated in the case of inadequate disease control from the above treatments. Three biologics have been licensed for the treatment of HS: adalimumab, secukinumab, and bimekizumab.
Licensed biologic |
Dosage |
More information |
|||||
Adalimumab (anti-TNF) |
|
|
|||||
Secukinumab |
|
|
|||||
Bimekizumab |
|
|
|||||
There is also limited data to support the use of infliximab, ustekinumab, and brodalumab in moderate to severe HS.
Therapeutics in phase II/III development include:
Surgery is an effective treatment that should be used in combination with lifestyle and medical intervention. Small nodules are ideally addressed early to prevent rupture of blocked follicles and the subsequent development of chronic, interconnecting sinus tracts.
It is usually safe to continue biological treatments whilst undergoing surgery, but specialist surgical opinion should be sought on an individual basis.
Procedural options are listed below:
Incision and drainage
Deroofing (unroofing)
Wide local excision
Radical excisional surgery
Risks of surgery include wound breakdown, numbness, several months of post-operative healing, and lesion recurrence.
Hair removal may be considered in non-inflammatory HS using laser therapy (eg, Ng:Yag, alexandrite, or diode lasers) or intense pulsed light therapy.
This may improve pain and slow disease progression by destroying the folliculo-sebaceous unit, thereby reducing follicular blockage and decreasing bacterial presence.
Hidradenitis suppurativa is a genetically driven, chronic scarring condition that can significantly impair quality of life if left untreated. Spontaneous remission or reduced inflammation may occur over time (eg, after menopause in women), although scarring persists. Early diagnosis and treatment are essential to minimise lasting damage.