Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Created 2008.
Although the majority of skin biopsies are successfully processed using routine formalin fixation and stained with haematoxylin and eosin (H&E), occasionally special stains and processing techniques are required for dermatopathological diagnosis and clinical management.
Routine H&E staining does not identify all tissue substances in certain inflammatory and neoplastic skin diseases it may be necessary to stain sections with a variety of different agents or perform different procedures.
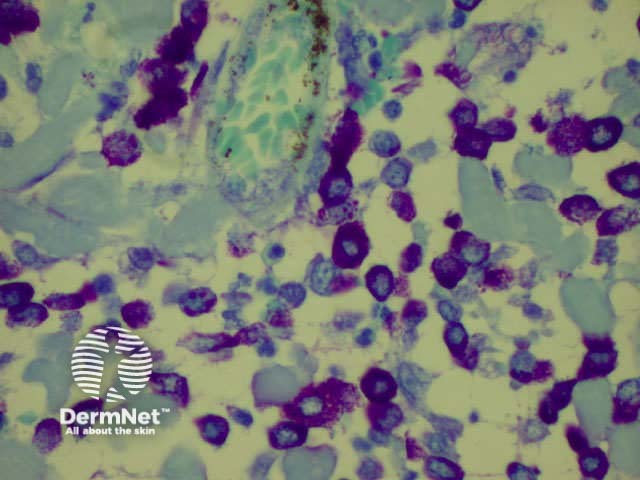
H&E of normal skin Mast cells (toluidine blue) 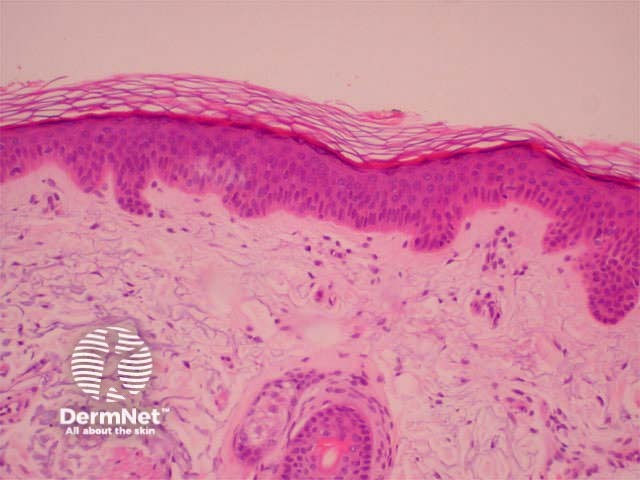
Various histochemical stains used to be used to identify cell types or specific substances in sections. These could be complex and fickle, and many have now been rendered largely obsolete in routine practice by the development of immunohistochemistry. Some histochemical stains are still very useful.
| Tissue material to be demonstrated | Stains used |
|---|---|
| Mucin (mucopolysaccharides) |
|
| Melanin |
|
| Iron (haemosiderin) |
|
| Calcium |
|
| Fibrin |
|
| Elastic fibres |
|
| Fat |
|
| Mast cells |
|
| Bacteria |
|
| Fungi |
|
At times during a surgical procedure, it is necessary to ask the pathologist for rapid diagnosis to plan treatment. This is almost always to allow assessment of the resection margins to check the completeness of excision in cases where the extent of the lesion is not easy to see clinically and when major reconstruction will be required at the time of surgery. Sometimes at critical anatomical sites, a special technique of frozen section control (Mohs surgery) may be used.
Except in very rare cases, it is not generally considered the good practice to make a primary diagnosis of malignancy on a frozen section. This is particularly true in the case of pigmented lesions, where the diagnosis, even on ideally processed paraffin sections, may be extremely difficult.
Frozen sections should be discussed with the pathologist prior to commencing surgery to ensure that both the pathologist and a technologist will be available at the required time.
The tissue is snap frozen t–20 t–70C in a cold liquid such as liquid nitrogen. The frozen tissue is solid enough to be sectioned with a microtome in a cryostat and the sections are then picked up on a glass slide ready for staining.
The sections are thicker than standard paraffin sections and the tissue quality is not as high. For this reason, tissue taken for a frozen section is always subsequently processed for paraffin sections to check the report, and occasionally features are found in these sections that were not apparent (or not present) in the frozen section.
Immunohistochemistry is a very powerful tool for identifying cells and substances in tissue sections by using the specificity of antigen-antibody binding. Commercially available antibodies are raised against particular cellular constituents and these, when applied in solution to a tissue section, will bind specifically to those constituents. The bound antibody is linked by a further step ta coloured indicator that can be seen down the microscope (‘staining’).
Despite the specificity of antigen-antibody binding, immunohistochemistry is not a specific science. Staining is in general reliable and reproducible, particularly using the commonest markers, for which a vast amount of experience is now available, but aberrant staining can and does occur (sometimes related to varying fixation). Therefore, a single positive or negative reaction should not overrule the basic histological findings in a case. Immunostaining should, in general, involve a panel of different antibodies to minimise the effect of a single aberrant result, and the panel requested should be designed to answer a particular question related to the differential diagnosis based on the H&E appearances.
The major uses of immunohistochemistry are to identify a poorly differentiated malignant tumour as a carcinoma, sarcoma, melanoma or lymphoma; to identify scanty tumour cells or tumour obscured by inflammation (for instance assessing the depth of a heavily inflamed melanoma); or for identifying a dense lymphoid infiltrate as lymphoma by demonstrating clonality of immunoglobulin light chain expression. Immunohistochemistry has little role in the diagnosis of inflammatory skin conditions.
The most commonly used antibodies are those raised against cytoskeletal proteins (‘intermediate filaments’) such as cytokeratin, desmin and actin, and markers of melanocytic lineage or differentiation. There are also numerous markers of lymphoid differentiation. Hundreds of markers are now available (of variable utility!). A selection of the most commonly used ones is given below. See Resources for a link ta comprehensive list.
| Antibody Target | Antibody | Identifies |
|---|---|---|
| Intermediate Filaments | Cytokeratin Desmin Actin |
Epithelial differentiation Muscle differentiation Muscle (much less specific) |
| Melanocytic differentiation | S100 HMB-45 Melan-A |
Sensitive, but non-specific; More specific, but not sensitive; More specific, sensitive |
| Lymphoid / Haematological Markers | Leucocyte common antigen CD3 CD4/CD8 CD20/CD79a CD30 CD68 Kappa/Lambda |
Most mature lymphocytes T cells T cell subsets B cells Large lymphoid cells Macrophages Immunoglobulin light chains |
Direct immunofluorescence staining is a specific type of immunohistochemistry, usually to detect the presence of immunoglobulins and complement in the skin. A batch of fluorescein isothiocyanate-labelled antibodies against IgG, IgM, IgA, fibrin and C3 is applied to frozen sections of fresh tissue and examined by fluorescence microscopy.
Characteristic staining patterns are seen in the immunobullous diseases, lupus erythematosus and vasculitis.
Circulating skin antibodies are detected by indirect immunofluorescence. In this technique, the patient's serum is incubated with squamous epithelium, and any antibodies that bind are identified by a fluorescein-labelled probe.
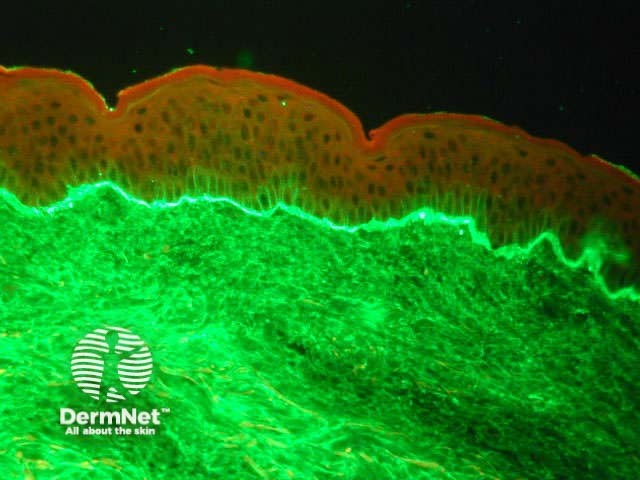
Normal skin Linear band of IgA at dermo-epidermal junction 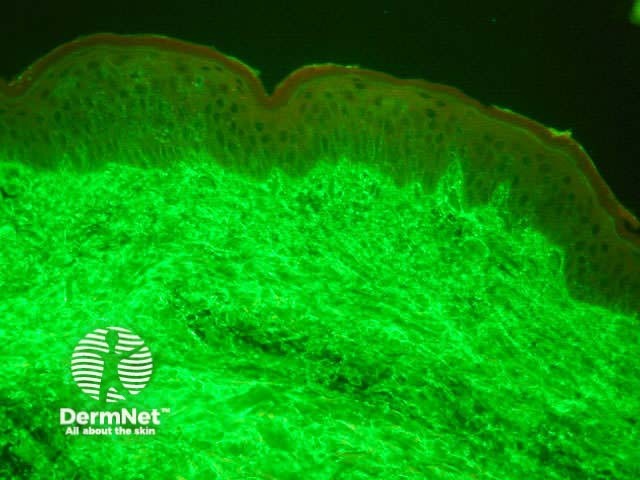
Electron microscopy (EM) is rarely required for routine diagnosis and management of skin diseases. Scanning EM is useful for diagnosing rare genetic hair shaft abnormalities. Scanning EM can be used to identify certain mineral deposits. Viral particles can be identified by EM, for example, to confirm a diagnosis of orf.
It remains a useful tool for research especially with the recent interest in photoageing, genetics, biotechnology and molecular biology.
EM is best performed when fresh tissue is fixed with glutaraldehyde. Suboptimal, but occasionally very useful, the examination can sometimes be made on appropriately treated formalin-fixed tissue if fresh material is not available. It can be combined with immunohistochemistry techniques for working up interesting cases, for the study of genetic and autoimmune bullous diseases. It is available from certain research laboratories in New Zealand.
In some cases identifying specific chromosomal alterations can confirm a diagnosis. This is particularly useful in cutaneous lymphomas where it can further classify the diagnosis, or help differentiate between systemic or primary cutaneous disease. The commonly used technique is fluorescent in situ hybridisation (FISH) where labelled DNA probes attach known areas on specific chromosomes. Equally, immunohistochemistry can be used to stain proteins gained or lost from these mutations.
The recent technique of laser-guided tissue microdissection is proving useful in identifying gene expression in localised areas of tissues seen under the microscope. DNA can be extracted from directly visualised clusters of cells. Single nucleotide polymorphism (SNP's) probes as markers of known mutations can then be used in conjunction with polymerase chain reaction (PCR) to identify whether a particular gene is being expressed in that area of the skin biopsy. This is largely a research technique currently.
Confocal laser scanning microscopy is used for obtaining high-resolution images and 3-D reconstructions in vivas it is applied directly to intact skin to view optical slices of the tissue. There is ongoing active research with this technique in dermatology and there may be a role for routine diagnostic use in the future. It has been used to image benign and malignant pigmented skin lesions, non-melanoma skin cancer, inflammatory skin conditions, and dynamic skin processes. There has also been some success with the mapping of indistinct pigmented lesions prior to surgery.
Examine a selection of pathologic reports. What special stains have been used, why, and what were the results in these cases?
Information for patients
See the DermNet bookstore.