Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Created 2008.
Skin lesions may be benign or malignant. Removal of lesions that involve the dermis or subcutaneous tissue requires surgical excision. Superficial skin lesions such as actinic keratoses, in situ squamous cell carcinoma and superficial basal cell carcinoma may be amenable to cryotherapy or photodynamic therapy (discussed in the physical therapy module) or topical therapy (described here).
Patients with sun damage, or who work outdoors, and all those with fair skin than tans poorly should be advised to protect their skin and eyes from the sun. Effective sun protection has been demonstrated to result in a reduction in actinic keratoses with time.
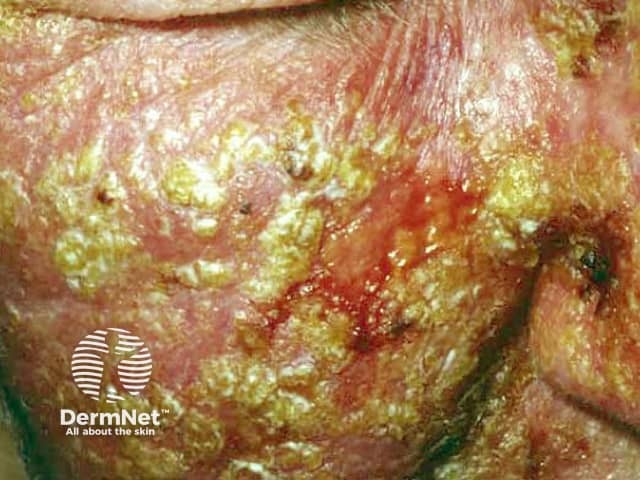
Topical 5-fluorouracil 5% cream (Efudix™) is a chemotherapeutic agent with partial Drug Tariff subsidy when prescribed by or on the recommendation of a dermatologist. It is a competitive antagonist for uracil in the formation of RNA and inhibits the incorporation of uracil into RNA. It is fairly effective in the management of actinic keratoses and in situ squamous cell carcinoma but is relatively ineffective for basal cell carcinoma. It is also sometimes used in combination with salicylic acid in the management of persistent and symptomatic viral warts in adults.
It works best on face and scalp, and is less effective on other areas. Pre-treatment with a topical retinoid enhances the effect of fluorouracil by peeling off the top layer of skin.
The optimal way to use fluorouracil is unknown. Most dermatologists recommend twice daily applications for 2 to 4 week courses, which may be repeated as necessary. To reduce the severity of adverse reactions, others recommend treatment for a few days, repeated as necessary, or twice weekly applications over several months.
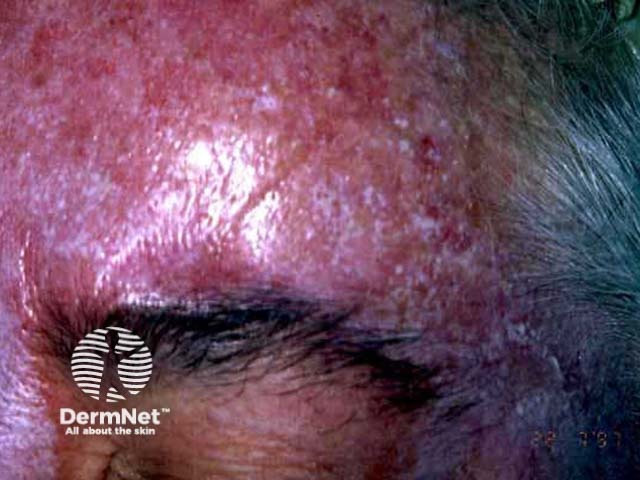
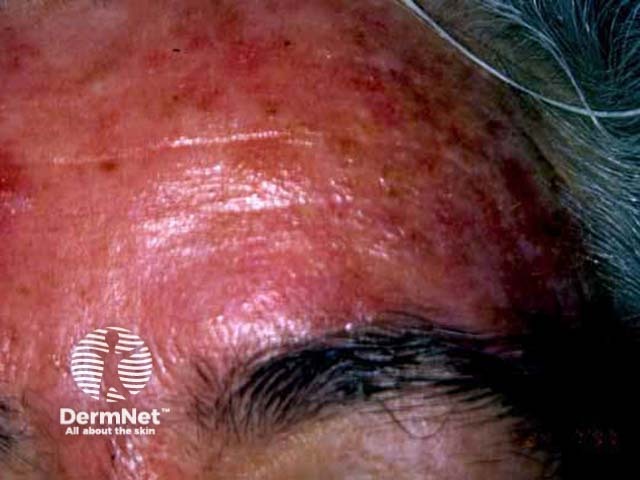
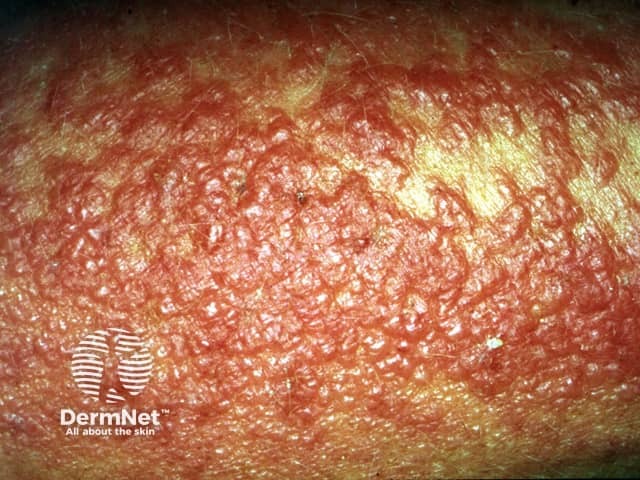
Fluorouracil is a cytotoxic destructive to dysplastic cells. Treated dysplastic lesions become red, scaly and tender over several days then erode or ulcerate with continued treatment. The more dysplastic the lesion, the more vigorous the response. Treatment should be discontinued at the eroding stage to allow re-epithelialisation over the next one to four weeks.
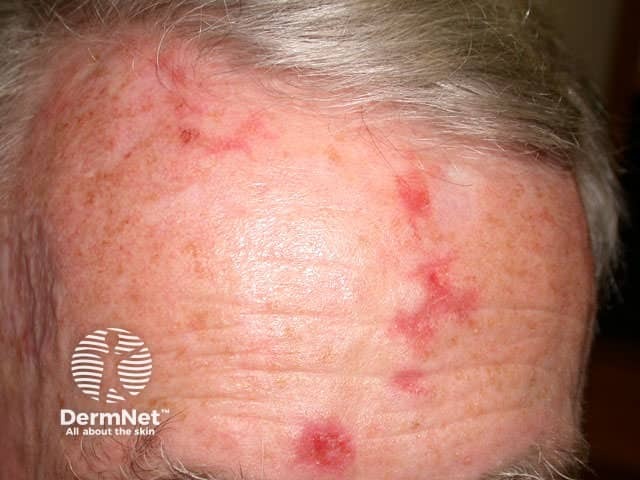
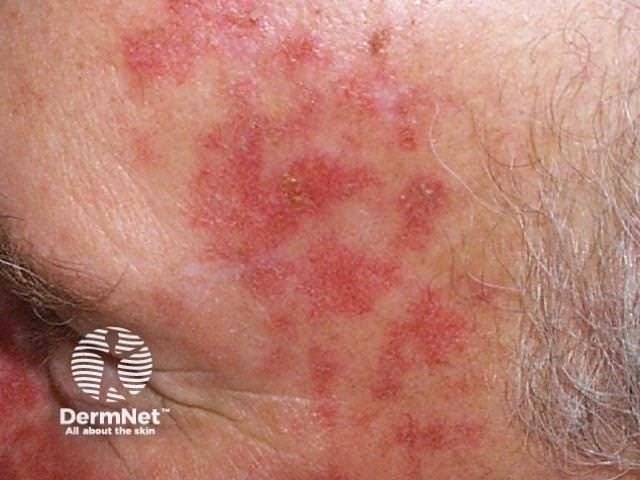
If treatment results in ulceration, healing may result in permanent scarring. This is most likely when in situ squamous cell carcinoma is affecting the lower leg, so careful supervision is necessary for lesions in this site and treatment should be interrupted if there is any erosion.
Fluorouracil may cause a diffuse phototoxic reaction in the areas of application so sun exposure should be avoided; normally treatment should be undertaken during the winter months.
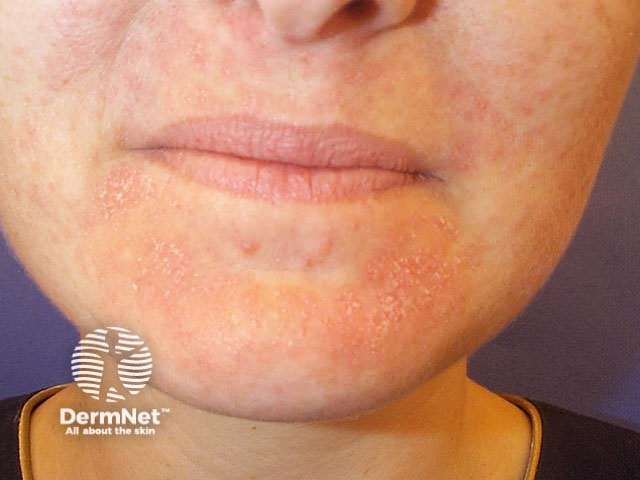
It is very irritant to mucous membranes so if actinic cheilitis is to be treated advise stopping it as soon as any soreness occurs (a few days).
Normal skin may be unaffected by the treatment, or may develop a contact irritant or allergic dermatitis. This may be treated by discontinuing the fluorouracil and applying emollients and topical steroid cream.
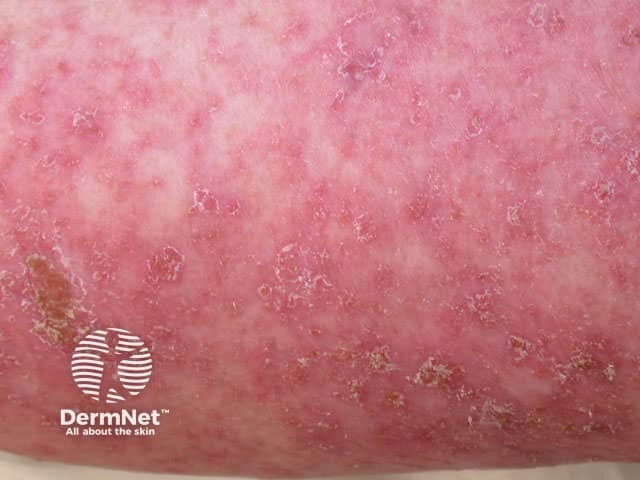
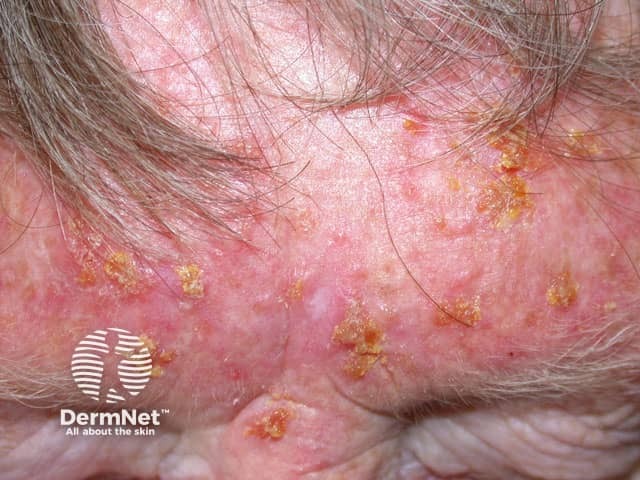
Imiquimod (Aldara™) is a an immune response modifier used to treat genital warts, actinic keratoses and superficial basal cell carcinomas (BCC)s. It may also be useful with other viral infections (e.g. plane warts, molluscum contagiosum) and tumours (e.g. in situ squamous cell carcinoma, nodular basal cell carcinoma).
Imiquimod binds to Toll-like receptor 7 on epidermal antigen presenting cells (Langerhans cells) and induces the innate and cell-mediated local immune response via the production of alpha interferon and other cytokines and infiltration of tumour destructive T-cell lymphocytes and macrophages.
The degree of inflammation is quite variable. The greater the inflammation, in general the shorter the time and number of applications required to eradicate the lesion. About 10% get little or no inflammatory response and treatment is then nearly always ineffective. On the whole, as with other topical treatments, facial lesions clear quicker than those elsewhere.
A course of treatment ranges from 4 to 16 weeks.
Mild reaction Moderate reaction Severe reaction Prior to treatment During treatment After treatment 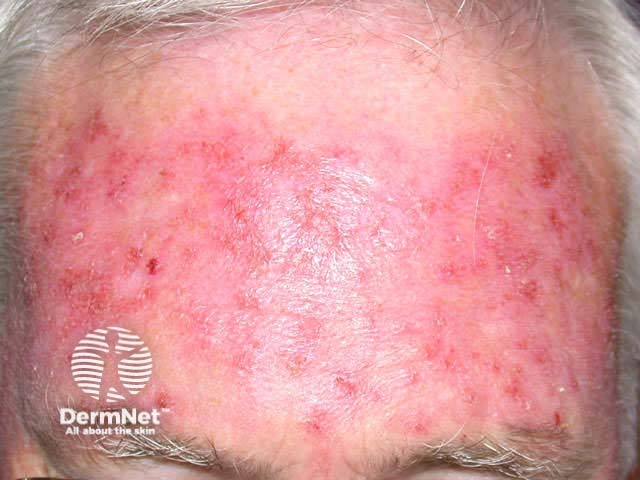
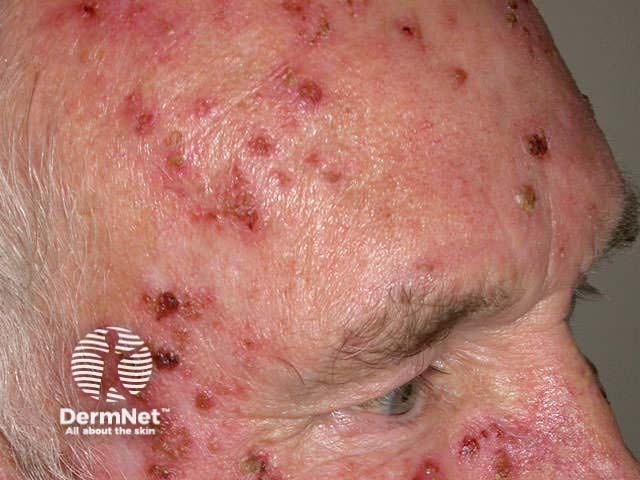
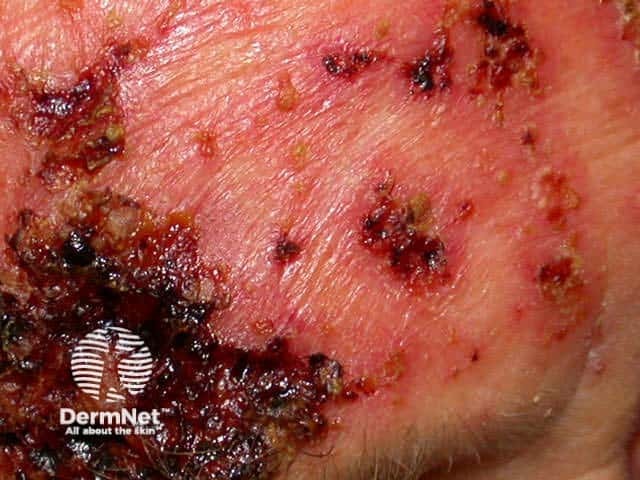
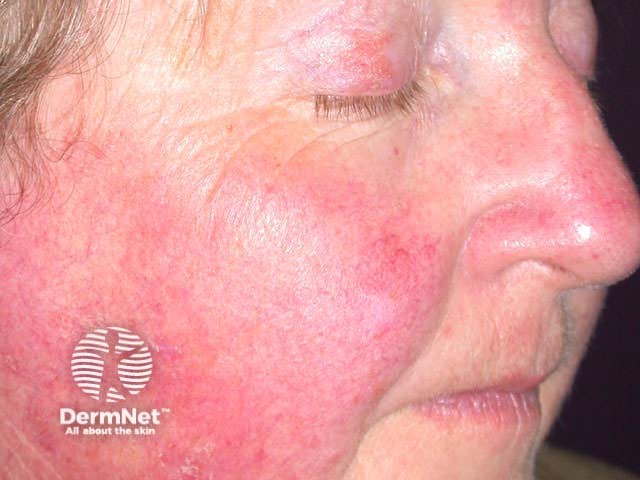
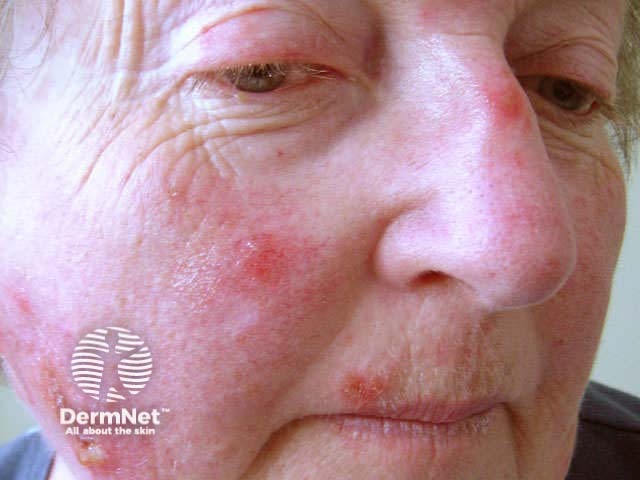
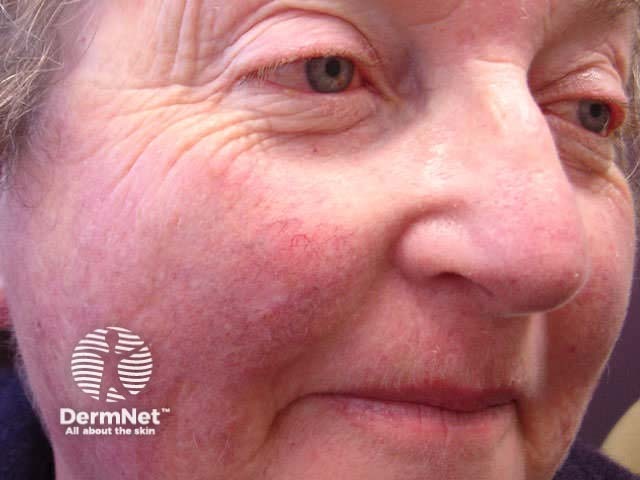
Excessive inflammatory reactions may cause erosions, ulceration and eventual scarring.
Occasional 'flu-like symptoms such as fever, fatigue, headache, nausea, diarrhoea and muscle pain may be treated with paracetamol. The course of treatment may need to be interrupted. Side effects should resolve within a few days of stopping treatment. They may also resolve with continuing treatment.
Observe for resistant or recurrent tumours. Occasionally, a nodular basal cell carcinoma may arise in an apparently successfully treated superficial basal cell carcinoma. The reason for this is uncertain.
Eroded lower lip Necrotic BCC reaction Hypopigmentation Nodular recurrent BCC 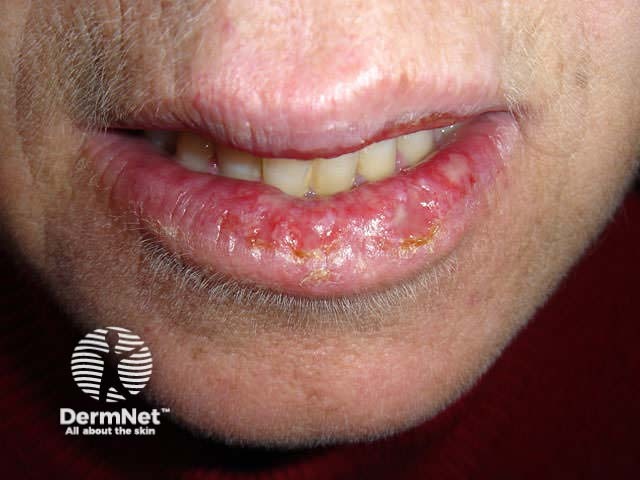
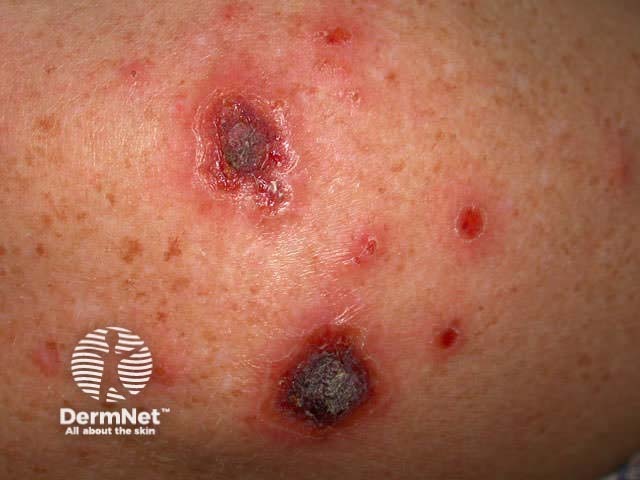
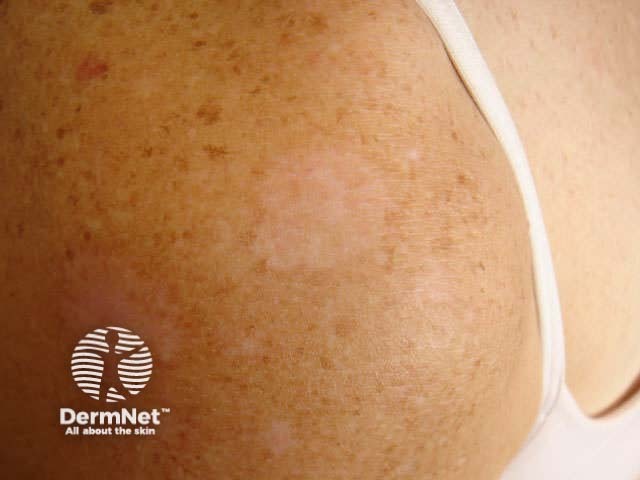
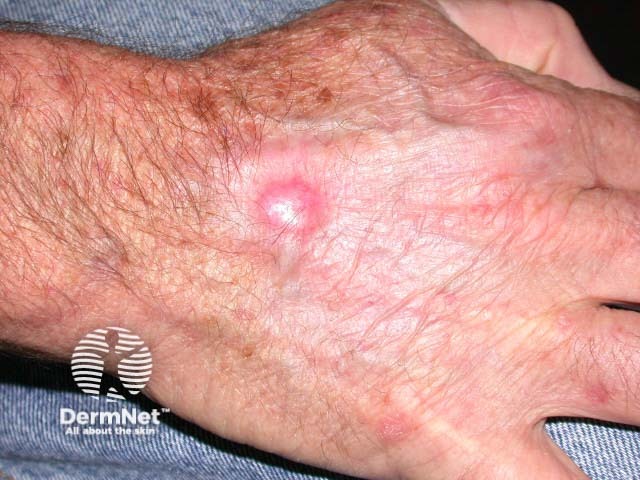
Ingenol mebutate is extracted from a common weed, Euphorbia peplus. It is approved in the USA for the treatment of actinic keratoses. It is available as 0.015% gel for face and scalp and 0.05% gel (Picato™) for trunk and limbs, applied once daily to individual lesions or an area of up to 5 x 5cm for 2-3 days. There is an inflammatory reaction followed by clearance or diminution in most lesions. Treatment can be repeated if necessary.
Diclofenac in hyaluran gel (Solaraze™) is available in several countries to treat actinic keratoses, but not yet in New Zealand (September 2008). It is applied twice daily to affected areas for about 12 weeks and is well tolerated. Lesions tend to improve for a few weeks after it has been discontinued, and then to slowly recur. It can be used again as necessary.
Topical non-steroidal anti-inflammatory preparations can sting broken skin and may cause irritant, allergic or phototoxic contact dermatitis. Large amounts may result in systemic effects including hypersensitivity, asthma and renal dysfunction.
Trichloracetic acid (TCA) is used as a peeling agent in various concentrations (15 to 80%). It is particularly effective at reducing blotchy pigmentation and may also improve fine lines and wrinkles (not deep furrows). It is not used to remove malignant skin lesions.
Higher concentrations result in partial thickness epidermal destruction and can also be used as spot treatment for seborrhoeic warts, viral warts and xanthelasma. Specific training is required in its use. Actinic keratoses improve but tend to recur within months.
TCA results in immediate discomfort as the skin surface is necrosed. Treated areas become red and swollen, then a scab forms that peels of in 7 to 10 days. TCA may cause permanent hypopigmentation and scarring.
Pre-treatment Immediately after application 48 hours later 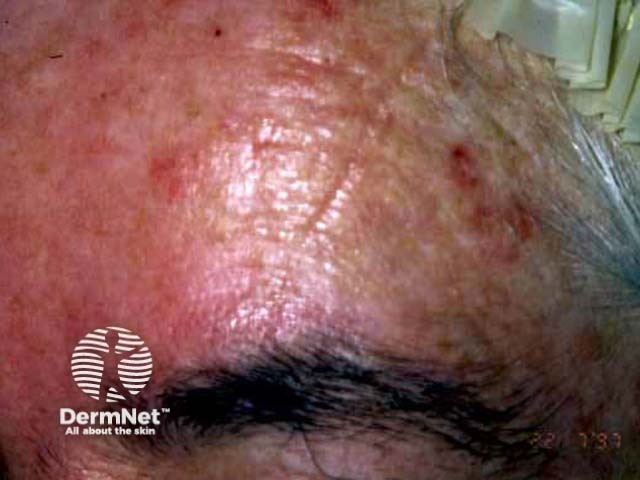
Tretinoin cream (Retin-A™), isotretinoin gel (Isotrex™) and adapalene (Differin™) gel/cream are prescription medicines for the treatment of acne. Tretinoin is also available in an emollient cream (Retinova™) to treat photoageing (fine lines, discolouration etc.) and is most often used on the face, the neck and the back of hands. It is not very effective at removing actinic keratoses and is inappropriate for invasive skin cancers.
Over-the-counter topical retinoids (retinol and retinaldehyde) are widely promoted to have anti-ageing properties. They peel off and lighten pigmented lesions (lentigines) and reduce fine surface wrinkles by preventing loss of collagen from, and stimulating new collagen formation in, the papillary dermis. They have a similar but less obvious effect on aged skin in non sun-exposed sites.
Topical retinoids frequently irritate and may increase the risk of sunburn. Excessive use results in redness and peeling. These effects can be minimised by using a minimal amount on alternate nights at first and if necessary applying a light oil-free moisturiser.
There is continuing controversy whether the topical use of tretinoin, isotretinoin and adapalene could cause birth deformities so they should not be prescribed in pregnancy. However dietary intake results in higher levels of detectable retinoids in serum than topical use.
Various topical agents applied at night or as superficial peels at a salon may improve the appearance of photoaged facial skin. In general, the ingredients do not penetrate deeply enough to eradicate actinic keratoses and are inappropriate in the management of skin cancer. They may be recommended as part of a general sun protection regimen; it is not known which of many possible options is the most effective and results are often disappointing. A so-called ‘serum’ is supposed to have a higher concentration of active ingredients than a cream. These products are often promoted with pseudoscience and outrageous claims of efficacy.
Ingredients may include:
As with almost any skin care products, contact irritant reactions are quite common (stinging, burning and dermatitis) and contact allergic dermatitis may occur less often. Most reactions are mild and settle spontaneously, but persistent dermatitis may require management with topical steroids and bland emollients. True allergy is most often due to a preservative, but has been reported from many ingredients.
The best protection from sun damage is to stay indoors. If outside, then protection from directly incident light can be obtained by seeking shade: trees, verandas, and umbrellas.
Clothing may be labelled with Ultraviolet Protection Factor ratings (UPF) according to Standard AS/NZS 4399:1996 Sun protective clothing – evaluation and classification. Standard AS/NZS 1067:2003 Sunglasses and fashion spectacles, specifies the physical properties sunglasses must have to protect the wearer from UV damage.
Daily and year-round use of sunscreens diminishes signs of photodamage and may reduce the number and severity of actinic keratoses.
Apply sunscreen to all uncovered skin liberally. Reapply after half an hour to ensure an even coating soaks into the skin, and then follow the instructions on the container. These generally advise reapplication 2-hourly and after bathing.
Sun Protection Factors (SPF) are a useful guide to the efficacy of a sunscreen. In New Zealand most manufacturers adhere to the requirements of a voluntary Australian and New Zealand Standard in which the labelled SPF can be up to 50+ (AS/NZS 2604:2012). The SPF is calculated from in-vivo testing, and is the ratio of the time for the skin to burn when exposed to solar simulating UVR with the sunscreen at a thickness of 2mg/cm2, compared without the sunscreen. Thus white skin that normally burns in 10 minutes will burn in 150 minutes (2 ½ hours) with SPF 15 sunscreen, however often it is reapplied. In general, sunscreens are much less effective than predicted by their SPF number as they are applied significantly less thickly than is tested and are rubbed, sweated or washed off.
The fern extract, Polypodium leucotomas, taken by mouth appears to have modest photoprotective effects and is available as a food supplement. Other oral antioxidants, including nicotinamide and carotenoids, may also be of benefit.
Sunscreens can act as chemical absorbers of a range of wavelengths of UVR (UVB and/or UVA) and as more broad spectrum physical reflectants of visible light (VL). Distinguishing specific ingredients can be tricky because of multiple chemical and trade names for the same substance (refer to TGA guide, referenced below).
| Drug Name | Absorption spectrum |
|---|---|
| Aminobenzoic acid | UVB |
| Butyl methoxydibenzoylmethane | UVAI |
| Cinoxate | UVB |
| Dioxybenzone | UVB, UVAII |
| Homosalate | UVB |
| Menthyl anthranilate | UVAII |
| Octocrylene | UVB |
| Octyl methoxycinnamate | UVB |
| Ethylhexyl salicylate | UVB |
| Oxybenzone | UVB, UVAII |
| Padimate O | UVB |
| Phenylbenzimidazole sulfonic acid | UVB |
| Sulisobenzone | UVB, UVAII |
| Titanium dioxide | UVB, UVA, VL |
| Triethanolamine salicylate | UVB |
| Zinc oxide | UVB, UVA, VL |
Sunscreen products may cause a variety of adverse effects.
Fluorouracil Fluorouracil 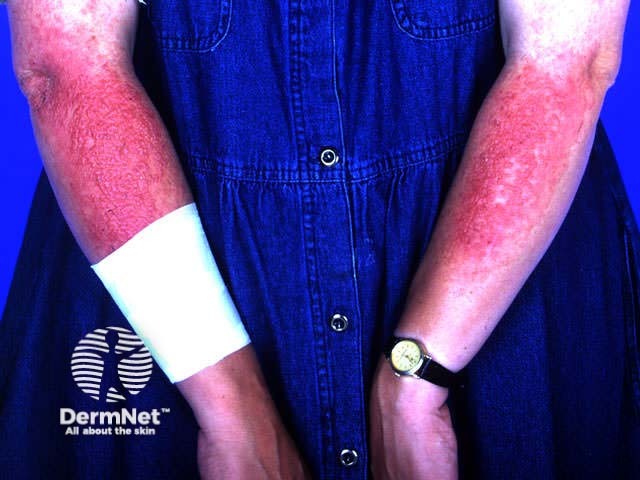
What is the effect of strict sun protection on vitamin D levels?
Information for patients
See the DermNet bookstore.