Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Haylee Boyens, Medical Student, University of Auckland, New Zealand, September 2014.
Introduction - plasma cells Introduction Demographics Causes Clinical features Diagnosis Treatment Prognosis Multiple myeloma
Plasma cells are white blood cells produced in the bone marrow and are found in the blood, the skin, and throughout the body. Their function is to make immunoglobulins (antibodies) to fight disease. They are an important part of the immune system.
A plasmacytoma is a tumour made up of abnormal plasma cells. It usually grows within the bone. When it grows in a site other than bone, it is called an extramedullary plasmacytoma.
There can be a single tumour (solitary plasmacytoma) or many tumours (multiple myeloma)
Plasmacytoma is rare. It affects middle aged and elderly people and is very rare under the age of 30 years. Cutaneous plasmacytoma is very rare.
It is not known what causes plasmacytoma. Radiation, industrial solvents and airborne toxins have been identified as possible risk factors.
Plasmactyoma may cause:
Extramedullary plasmacytoma can occur at any site, but 80–90% of extramedullary plasmacytomas are in the head and neck area, particularly within the upper airways and oral cavity. Symptoms may include:
Cutaneous/skin involvement is very rare, accounting for 2–4% of all extramedullary plasmacytomas. They typically present as red nodules or dome shaped plaques, and may ulcerate.
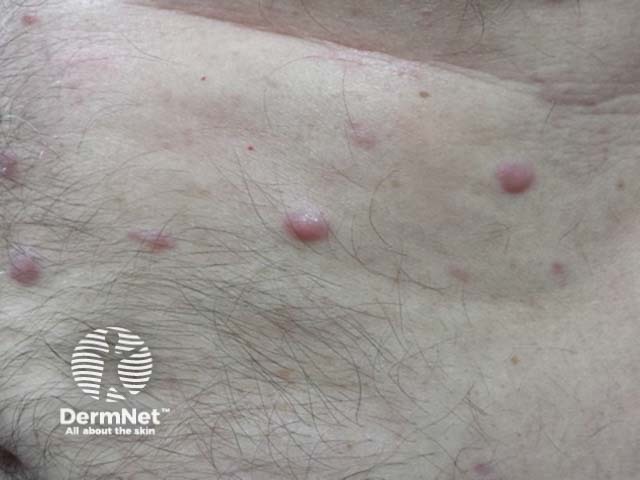
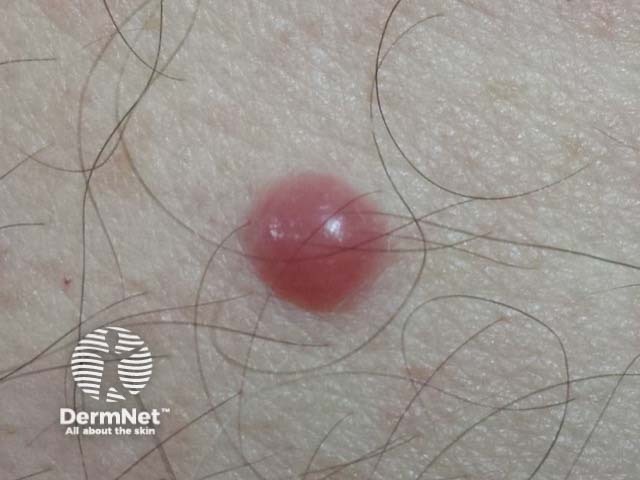
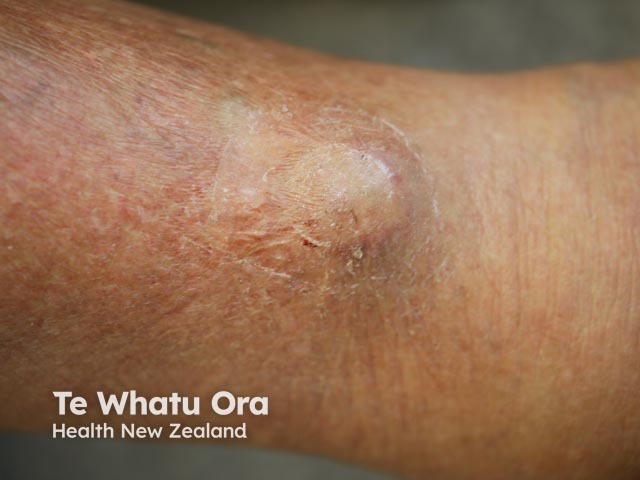
Cutaneous plasmacytoma
Plasmacytoma is diagnosed by a tissue biopsy or bone marrow biopsy. This shows invasion of the bone or tissue by monoclonal (identical) plasma cells (see cutaneous plasmacytoma pathology). To make a diagnosis of solitary plasmacytoma (of bone or extramedullary site), other plasma cell tumours or multiple myeloma must be excluded.
Solitary plasmacytoma is treated by radiotherapy or surgery.
The prognosis for plasmacytoma depends on whether the lesions are solitary or a sign of multiple myeloma.
Multiple myeloma may later develop in patients with solitary bone plasmacytoma (65–84% at 10 years) or extramedullary sites (11–30% at 10 years).
Cutaneous involvement in patients with known multiple myeloma usually indicates a poor prognosis regardless of treatment.
Normally, plasma cells account for <5% of bone marrow. Plasma cell cancers occur when there is uncontrolled proliferation of these cells. There are two forms:
Plasma cell leukaemia is rare and aggressive. It can be primary (the first sign of disease) or secondary to advanced multiple myeloma.
Approximately 10–15% of patients presenting with a solitary extramedullary plasmacytoma and 50–60% of patients presenting with solitary plasmacytoma of the bone will ultimately develop multiple myeloma.
Extramedullary plasmacytomas are seen in around 7% of patients who have multiple myeloma at the time of diagnosis, and a further 6% of patients will go on to develop extramedullary plasmacytomas after being diagnosed with multiple myeloma.
Multiple myeloma presents with the following signs and symptoms.
The following tests are used to detect and monitor multiple myeloma.
If multiple myeloma is present, it may be treated by:
Treatments may be used individually or in combination, depending on the case details and risk profile.
Predicting prognosis in multiple myeloma is not straightforward, as some patients remain asymptomatic for a number of years while for others the disease is rapidly progressive.
These estimates of prognosis may be adjusted depending on availability of and response to treatment, patient characteristics, and tumour genetics. Some tumour mutations are associated with more aggressive forms of disease which may shorten survival time.
Patients are categorised as high, medium or standard risk using cytogenetic testing, which is done on the tumour cells to identify genetic abnormalities which may contribute to a more aggressive disease pathway.
The standard and medium risk groups have an estimated median survival of 8–10 years. Patients in whom certain genetic abnormalities are detected are likely to have a shorter survival time, as are patients in whom disease is advanced.