Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Introduction Clinical features Causes Investigations Treatment
There are two forms of heparin-induced thrombocytopenia, type 1 and type II. Heparin-induced thrombocytopenia type I is harmless, causing a mild and transient reduction in platelet count.
Heparin-induced thrombocytopenia type II (HIT) is a more serious autoimmune disease causing a low platelet count and paradoxical blood clotting.
Heparin can also give rise to other cutaneous reactions, including:
Two-thirds of people with heparin-induced thrombocytopenia develop a reduction in platelet count without symptoms.
However, approximately 30% of patients with heparin-induced thrombocytopenia develop thrombosis (heparin-induced thrombocytopenia with thrombosis or HITT). Thrombosis can occur in veins, arteries or in the small vessels in the skin, in the brain (causing stroke), in the legs (causing deep vein thrombosis), in the lungs (pulmonary embolus), in the heart (myocardial infarction), or in fingers and toes (digital necrosis). The blood clots block the affected vessel causing necrosis.
Skin necrosis manifests as sharply demarcated tender red or purple patches (purpura) which develop black centres and may lead to dry gangrene. A net-like or branching, retiform, pattern of purpura may also be seen. Necrosis may occur at the site of subcutaneous injection or infusion of heparin, or at distant sites.
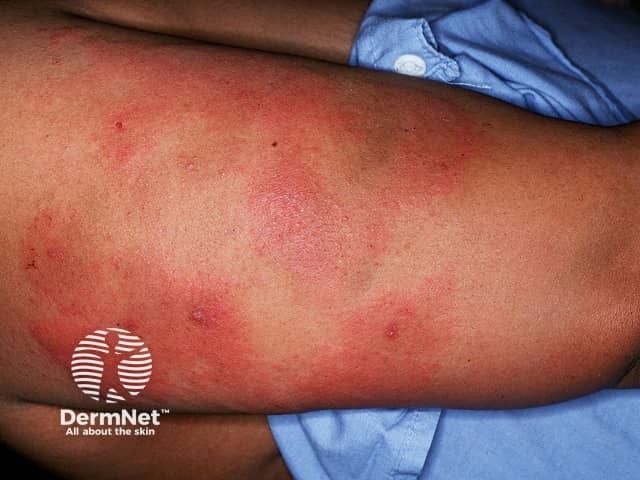
Purpura due to heparin-induced thrombocytopenia
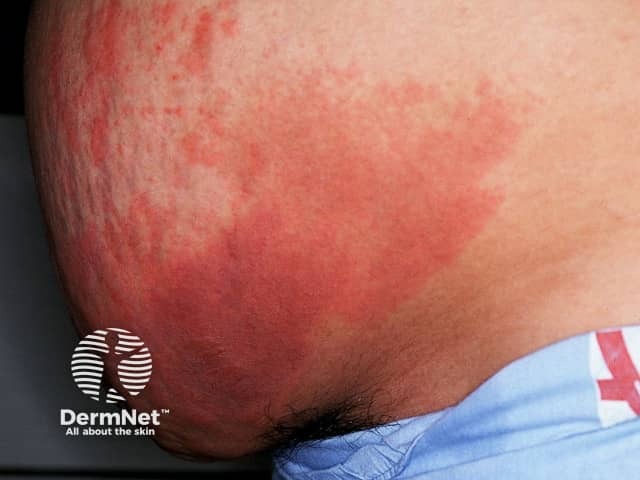
Purpura due to heparin-induced thrombocytopenia
Heparin is a widely used natural anticoagulant or 'blood thinner'. It works by activating antithrombin, which in turn inactivates thrombin and prevents clot formation.
Heparin-induced thrombocytopenia type II is due to an antibody that binds to complexes of heparin and platelet factor 4. The Fc binding protein on the antibody complex matches up with Fc receptors on the platelets, activating them. Platelet aggregation and consumption follows. Platelet levels reduce from pre-treatment levels. If platelet aggregation is severe enough it causes the formation of a 'white clot' which results in a complete blockage of the affected artery or vein. The specific symptoms depend on the organ to which the blood supply is blocked.
Antibodies take time to develop in autoimmune diseases. Heparin-induced thrombocytopenia usually manifests 5–10 days after starting treatment. Patients who develop heparin-induced thrombocytopenia within hours of starting treatment have likely been exposed and thus sensitised to heparin previously.
Unfractionated heparin is more likely to cause heparin-induced thrombocytopenia than fractionated heparin (also called low molecular weight heparin).
Patients on heparin should have their blood count measured regularly to check platelet levels. Heparin-induced thrombocytopenia may be diagnosed if there is a reduction compared to pretreatment levels, even when the absolute platelet level is still within the normal range.
Functional tests to detect heparin-induced thrombocytopenia include:
Immunoassays to detect antibodies to heparin-platelet factor 4 are more sensitive but less specific compared to functional tests.
Skin biopsy of cutaneous necrosis can help in diagnosis. In heparin-induced thrombocytopenia, there are blocked arteries, veins and small blood vessels without inflammation. Histology cannot distinguish white clots made up of platelet plugs from red fibrin clots.
Heparin injections or infusions should be stopped.
Alternative anticoagulation may be required to prevent life-threatening thrombosis. This is usually with factor Xa inhibitors such as danaparoid, or direct thrombin inhibitors such as bivalirudin or dabigatran.
There have been several cases of warfarin-induced skin necrosis when warfarin monotherapy has been used in patients with heparin-induced thrombocytopenia. This is thought to be because warfarin reduces protein C activity. Therefore warfarin alone is not recommended in patients with active heparin-induced thrombocytopenia.
Re-exposure to heparin is not recommended during the acute phase of heparin-induced thrombocytopenia and the post-acute period while there are still circulating antibodies. If heparin-induced thrombocytopenia occurred a long time previously, all antibodies may have cleared and it may be safe to use heparin again. However recurrent heparin-induced thrombocytopenia has been described in some patients.
Advice on treatment and re-exposure should be sought from a haematologist.