Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Vanessa Ngan, Staff Writer, 2003. Updated by Dr Jannet Gomez, Postgraduate Student in Clinical Dermatology, Queen Mary University London, United Kingdom; Chief Editor: Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand, February 2016.
Introduction
Causes
Pathophysiology
Clinical features
Diagnosis
Treatment
Outcome
Necrotising fasciitis is a very serious bacterial infection of the soft tissue and fascia. The bacteria multiply and release toxins and enzymes that result in thrombosis in the blood vessels. The result is the destruction of the soft tissues and fascia.
The main types of necrotising fasciitis are:
Bacteria causing type 1 necrotising fasciitis include Staphylococcus aureus, Haemophilus, Vibrio and several other aerobic and anaerobic strains (Escherichia coli, Bacteroides fragilis). It is usually seen in older people or in patients affected by diabetes mellitus or other conditions.
Type II necrotising fasciitis has been sensationalised in the media and is commonly referred to as a flesh-eating disease. It affects all age groups. Healthy people are also prone to infection with this group.
Type III necrotising fasciitis is caused by Clostridium perfringens or less commonly Clostridium septicum. It usually follows significant injury or surgery and results in gas under the skin: this makes a crackling sound called crepitus. People who inject “black tar” heroin subcutaneously can also be infected with clostridia and develop necrotising fasciitis.
Necrotising fasciitis due to marine organisms is usually due to contamination of wounds by seawater, cuts by fish fins or stingers, or consumption of raw seafood. It occurs more commonly in patients with liver disorders. These infections can be very serious and can be fatal if not attended within 48 hours.
Fungal necrotising fasciitis complicates traumatic wounds in immunocompromised people.
Other terms used for necrotising fasciitis include haemolytic streptococcal gangrene, Meleney ulcer, acute dermal gangrene, hospital gangrene, suppurative fasciitis, and synergistic necrotising cellulitis.
Necrotising fasciitis affecting perineal, genital, and perianal regions is known as Fournier gangrene. This has a particularly high death rate ranging from 15% to 50%.
Necrotising fasciitis may occur in anyone, with almost half of all known cases of streptococcal necrotising fasciitis occurring in young and previously healthy individuals. The disease may occur if the right set of conditions is present, these include:
Risks for necrotising fasciitis include:
The infection starts in the superficial fascia. Enzymes and proteins released by the responsible micro-organisms cause necrosis of fascial layers. Horizontal spread of infection may not be clinically apparent on the skin surface and hence diagnosis may be delayed. The infection then spreads vertically up into the skin and down into deeper structures. Thrombosis occludes the arteries and veins leading to ischaemia and necrosis of the tissues.
Streptococci produce:
Aerobic and anaerobic bacteria produce hydrogen, nitrogen, and hydrogen sulfide gases that destroy hyaluronic acid enabling the spread of infection.
Signs and symptoms vary between individuals but often some or all of the following are present.
By about days 4–5, the patient is very ill with dangerously low blood pressure and high temperature. The infection has spread into the bloodstream and the body goes into toxic shock. The patient may have altered levels of consciousness.
Metastatic abscesses can develop in liver, lung, spleen, brain, pericardium, and rarely, in the skin.
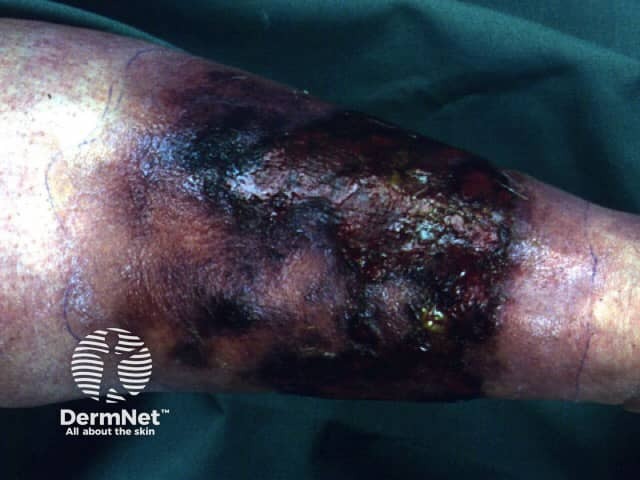
Necrotising fasciitis
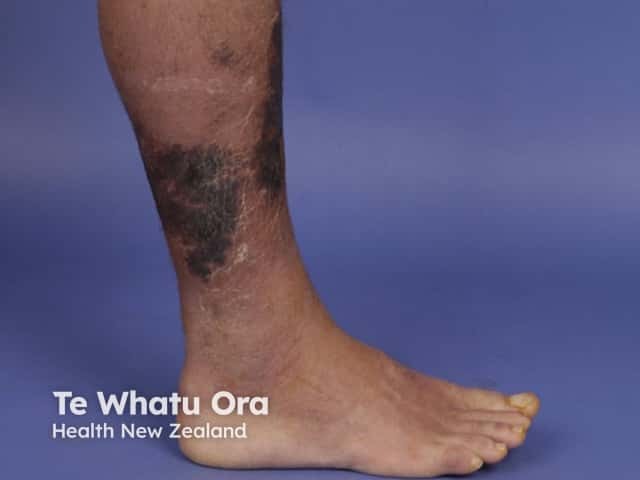
Necrotising fasciitis
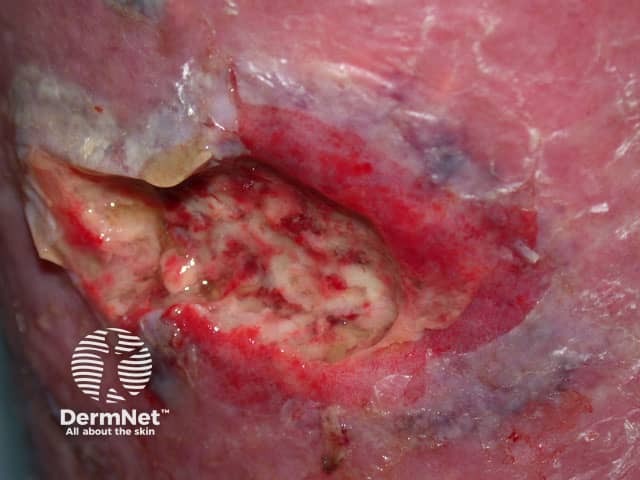
Necrotising fasciitis
See more images of necrotising fasciitis.
A thorough history and clinical examination are crucial in arriving at the diagnosis of necrotising fasciitis. Special care should be taken when examining immunocompromised patients, as the presentation of symptoms/signs may be atypical.
Blood culture, deep tissue biopsy [see Necrotising fasciitis pathology], and Gram stain help in identifying the culprit organism(s) and guide the choice of antibiotic. If Staphylococcus aureus is detected, MRSA sensitivity test should be done. Blood cultures are usually negative for clostridial species.
Fungal culture should be performed in immunocompromised and trauma patients.
X-ray, CT scan, and MRI identify areas of fluid collection, inflammation and gas within the soft tissues.
The Laboratory Risk Indicator for Necrotising Fasciitis (LRINEC) is a tool that aids in distinguishing necrotising fasciitis from other tissue infections based on six parameters. A score of ≥ 6 favours necrotising fasciitis. This test is not appropriate for all cases and is not completely reliable.
CRP (mg/dL) ≥15:
WBC count (109/L)
Haemoglobin (g/L):
Serum sodium (mmol/L) <135:
Creatinine (umol/L) >141:
Glucose (mmol/L) >10:
Once the diagnosis of necrotising fasciitis is confirmed, treatment should be initiated without delay.
Immediate surgical debridement improves survival and avoids complications of necrotising fasciitis. All infected tissue should be cleared away using adequate excision. Repeated debridements are carried out for a few days.
When the acute infection has subsided, the wound should be closed with skin grafting if required. Vacuum-assisted wound closing devices may be useful to heal a persistent ulcer.
Prompt diagnosis and treatment are essential to reduce the risk of death and disfigurement from necrotising fasciitis.
If diagnosed and treated early, most patients will survive necrotising fasciitis with minimal scarring. If there is significant tissue loss, later skin grafting will be necessary and in some patients amputation of limbs is required to prevent death.
Up to 25% of patients will die from necrotising fasciitis, due to complications such as renal failure and septicaemia (blood poisoning) and multiorgan failure.