Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Dr Eugene Tan, Dermatology Registrar, Waikato Hospital, Hamilton, New Zealand, 2009.
DermNet Update June 2021
Introduction
Causes
Demographics
Clinical features
Cutaneous features
Diagnosis
Treatment
Outcome
Whipple disease is a very rare bacterial infection that can affect many organ systems of the body. The disease is named after George Hoyt Whipple who described a case in 1907, but it was not until 1961 that an infection was recognised as the cause.
The cause of Whipple disease is the ubiquitous bacterium Tropheryma whipplei (T. whipplei), a gram positive rod. T. whipplei is widespread; it has been detected in soil, sewage water, and faecal material. It is an intracellular bacterium that infects macrophages causing secretion of interleukin (IL)-16, macrophage apoptosis, and loss of mitochondrial membrane potential. The organisms characteristically accumulate in macrophages in the duodenal mucosa. It is spread via the faecal-oral route.
Whipple disease is typically described in Caucasian men with a mean age of 50 years at the time of diagnosis. It is very rare with a global incidence estimated to be 12 cases per year. The prevalence is highest among sewage plant workers, farmers, and agricultural workers, or those with poor hygiene habits such as the homeless.
In some studies, up to 35% of unaffected healthy individuals carry T. whipplei in their intestines. Not all infected individuals have symptoms. The symptoms of Whipple disease are related to the immune system and its interaction with the bacterium.
Specific defects in the immune system may allow T. whipplei to disseminate widely throughout the body and cause disease.
The four classic clinical features of Whipple disease are:
However, Whipple disease can affect any organ system and has a broad spectrum of clinical features. Chronic localised forms of Whipple disease are being increasingly identified.
Clinical features of Whipple disease |
|
|---|---|
Central Nervous System |
|
Musculoskeletal system |
|
Cardiovascular system |
|
Non-specific symptoms |
|
Skin signs in Whipple disease are very rare. They may be due to malnutrition or to an immune reaction to the infection.
Malnutrition-related skin signs are due to malabsorption and diarrhoea.
Inflammatory skin changes are due to the immune response towards T. whipplei. They may resemble:
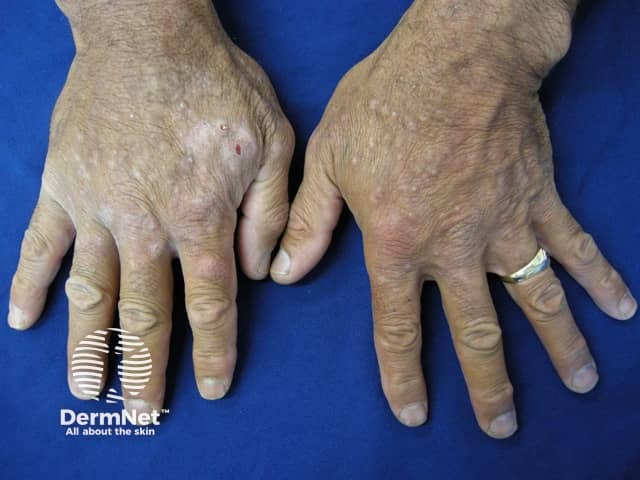
Nodules
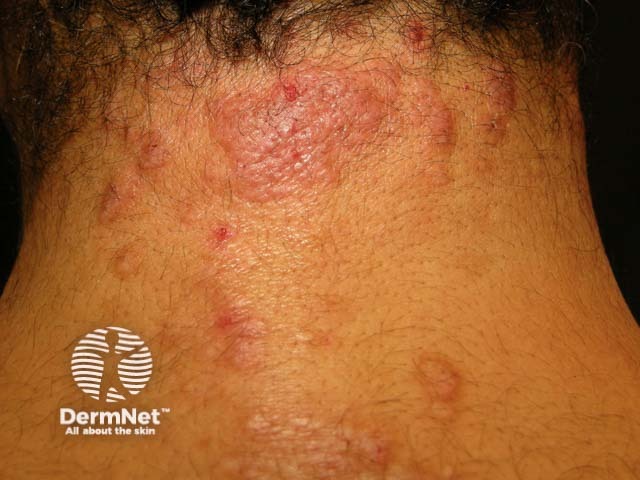
Eczematous plaques
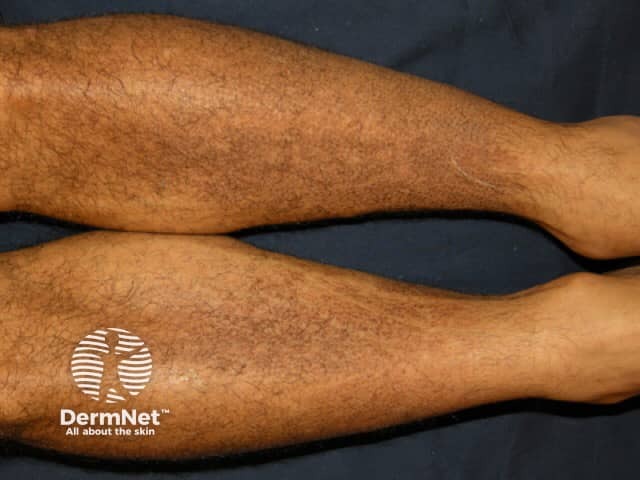
Hyperpigmented papules
The nonspecific symptoms and signs of Whipple disease together with its rarity make it a very difficult disease to diagnose.
Tissue biopsy is often necessary. The following tests may detect T. whipplei within the tissue:
Infected individuals often need high doses of antibiotics in case the brain is infected by T. whipplei. An induction regime with intravenous ceftriaxone 2 g once daily is given for two weeks.
Endocarditis and CNS involvement require a longer course of ceftriaxone or penicillin G.
Longterm antibiotics such as trimethoprim+sulphamethoxazole should then be continued for up to one year to ensure complete clearance of T. whipplei. Followup upper GIT endoscopy is recommended before ceasing treatment
Before the recognition of a bacterial cause and the use of antibiotics, Whipple disease was invariably fatal.
Treatment can be complicated by a Jarisch-Herxheimer reaction in the first few hours or the immune reconstitution inflammatory syndrome (IRIS) in the first two weeks.
After treatment, a 4-5 year (or longer) period of latency may ensue before recurrence in 10-15% if the infection is not completely eradicated particularly in patients with CNS involvement.