Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Autoimmune/autoinflammatory
Author: Hon A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand, 2001. Updated in December 2018.
Introduction
Uses
How it works
Side effects
Combined treatment
Calcipotriol is a vitamin-D derivative, about 1% as powerful as the natural hormone calcitriol (also known as 1,25 dihydroxycholecalciferol).
Known as calcipotriene (Dovonex®) in the United States, calcipotriol is available as an ointment, cream and scalp solution at a concentration of 50µg/g. In New Zealand, it is called Daivonex®, and, from 1 July 2016, only the ointment is marketed. Calcipotriol is not available in tablet or injection form.
Calcipotriol is also available in combination with betamethasone propionate as a gel or ointment (Daivobet®) and foam (Enstilar®).
Calcipotriol is a prescription medication used mainly for psoriasis. It is moderately or very effective for about 80% of patients. This means the patches become less scaly and thick, but red patches often persist despite continued treatment.
It should be applied to the affected patches of psoriasis twice daily and can be used safely long term. The ointment is used on trunk and limbs. The cream is more often used in the body folds or on the face and ears. The scalp solution is used in hairy areas.
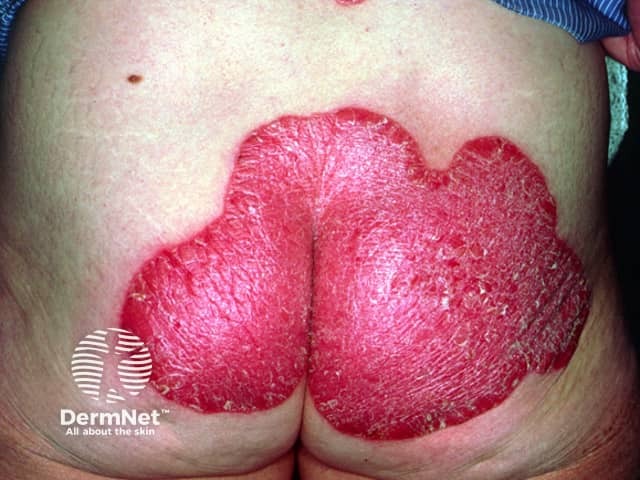
Psoriasis pre-calcipotriol
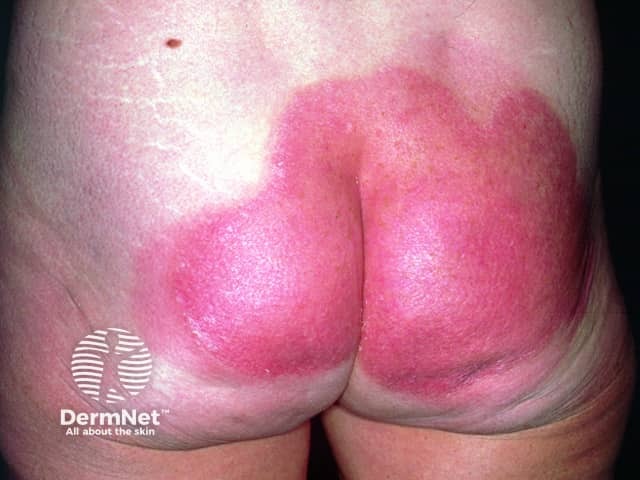
Psoriasis post-calcipotriol (after 6 weeks of treatment with calcipotriol ointment)
Calcipotriol is sometimes also helpful in the following skin conditions:
Calcipotriol acts like vitamin D. It is antiproliferative, reducing the abnormal proliferation of keratinocytes that occurs in psoriasis, and it induces cell differentiation, normalising epidermal growth.
Topical calcipotriol is usually well tolerated. Side effects are more common with the ointment formulation than with the cream (but the ointment is more effective on plaque psoriasis).
It is essential to prevent pets from ingesting calcipotriol - 5 grams of a 50µg/g formulation may be enough to produce fatal toxicity in a small dog. Keep tubes out of reach, wash hands thoroughly after application, and do not allow pets to lick areas of application.
Calcipotriol is often used with other treatments for psoriasis with good effect.
Calcipotriol may reduce the skin thinning effect of topical steroids.
Salicylic acid is used to remove scale but as it deactivates calcipotriol, it should not be applied at the same time. If combined use is required, apply them at a different time of day.
Ultraviolet radiation (UVR) deactivates calcipotriol, which could also act as a sunscreen, so the calcipotriol should be applied after UVR and not beforehand.
Some patients find calcipotriol more irritating when they are on acitretin because acitretin thins the skin and make it more sensitive. Use moisturisers and emollients regularly.
No special precautions are necessary with the combination of calcipotriol with methotrexate.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).