Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Autoimmune/autoinflammatory
Author: Anoma Ranaweera, Medical Writer, Auckland, New Zealand. DermNet Editor in Chief: Adjunct A/Prof Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand. Copy edited by Gus Mitchell. October 2019.
Introduction - psoriasis
Introduction
How it works
How to use
Contraindications
Warnings and precautions
Use in specific populations
Potential drug interactions
Side effects
Psoriasis is an autoimmune skin disorder characterised by circumscribed, red, scaly plaques.
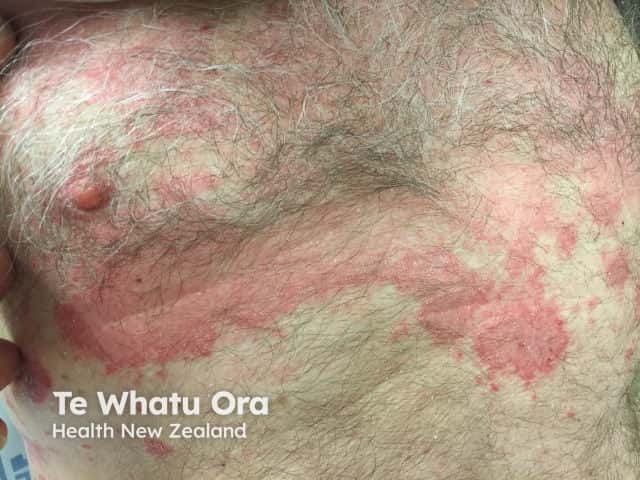
Chronic plaque psoriasis
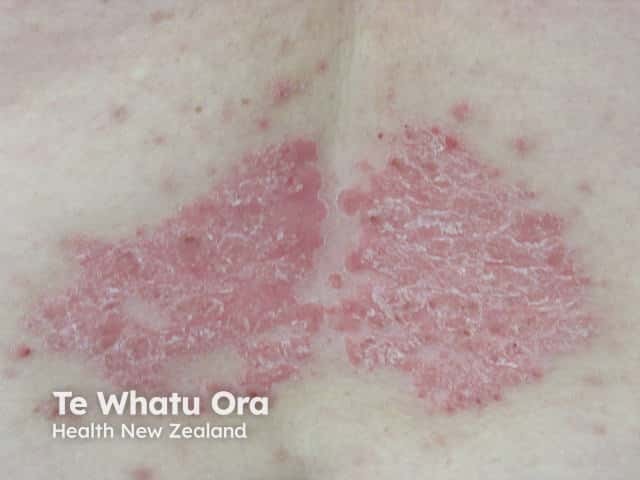
Chronic plaque psoriasis
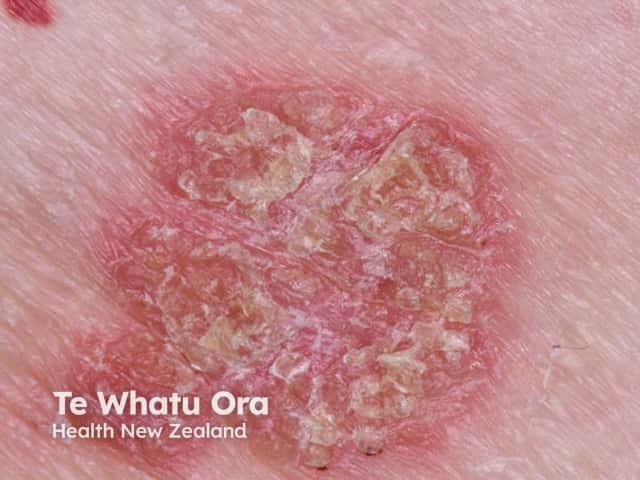
Chronic plaque psoriasis
Calcipotriol/betamethasone dipropionate foam (trade name Enstilar®) is a topical prescription medicine for psoriasis in adults 18 years and older. Enstilar is an alcohol-free foam formulation of the two drugs in a pressurised spray; this permits application across large areas [1,2].
The United States of America, the European Union, Canada, Australia, New Zealand, and other countries have approved Enstilar foam (calcipotriol/betamethasone dipropionate 50µg/g/0.5mg/g) for the treatment of plaque psoriasis.
The combination of calcipotriol and betamethasone dipropionate is also available as a gel, an ointment, and a cream (trade name Daivobet®, Dovobet®, and others).
The mechanism of action of the ingredients has been studied independently as well as in combination [1,3].
Calcipotriol hydrate is a synthetic vitamin D analogue.
Betamethasone dipropionate is a potent synthetic topical corticosteroid.
The foam formulation combines the pharmacological effects of calcipotriol and betamethasone dipropionate [2].
Each gram of foam contains 52.2 mcg calcipotriol hydrate (equivalent to 50 mcg of calcipotriol) and 0.643 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone) [1,4].
No more than 60 g of calcipotriol/betamethasone foam should be used in every four days.
Calcipotriol/betamethasone dipropionate foam is indicated for plaque psoriasis. The main contraindication to calcipotriol/betamethasone dipropionate foam is hypersensitivity to the active substance or to any of the excipients [1,5].
It should not be used for acute types of psoriasis such as generalised pustular psoriasis or erythrodermic psoriasis.
Calcipotriol/betamethasone dipropionate foam should also not be used for skin affected with:
Before using calcipotriol/betamethasone foam, patients should inform treating physicians in the following circumstances:
The propellants in calcipotriol/betamethasone dipropionate foam are flammable. Patients are instructed to avoid fire, flame, and smoking during and immediately following application. Keep the foam in a cool dry place, store below 25°C, and store away from sunlight. Do not expose the container to temperatures above 50°C as it is pressurised and extremely flammable [1,3].
Hypercalcaemia and hypercalciuria have been observed with the use of calcipotriol/betamethasone dipropionate foam and is most likely to occur if recommended doses are exceeded [1].
If hypercalcaemia or hypercalciuria develops, treatment should be discontinued until parameters of calcium metabolism have normalised.
The incidence of hypercalcaemia and hypercalciuria following calcipotriol/betamethasone dipropionate foam treatment of more than 4 weeks has not been evaluated.
Systemic absorption of topical corticosteroids, such as betamethasone dipropionate, can rarely produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency during treatment or upon withdrawal of the topical corticosteroid. Factors that predispose a patient to HPA axis suppression include treatment to large surface areas, prolonged use of occlusive dressings, altered skin barrier, liver failure, and young age [1].
Systemic effects of topical corticosteroids may also include Cushing syndrome, hyperglycaemia, and glucosuria.
Local side effects from topical corticosteroids include:
Misdiagnosis of fungal infection can lead to tinea incognito.
Allergy to topical corticosteroids may rarely result in allergic contact dermatitis, which has also been reported to occur with topical calcipotriol [1].
In clinical trials, no overall differences in calcipotriol/betamethasone foam exposure, safety, or effectiveness have been observed between older and younger subjects. In these trials, however, the number of subjects aged 65 years and older was not sufficient to determine whether they responded differently from younger subjects [1,3].
The safety and efficacy of calcipotriol/betamethasone foam in paediatric patients less than 18 years of age have not yet been established.
Because of a higher ratio of skin surface area to body mass, children under the age of 12 years are at particular risk of systemic adverse effects when they are treated with topical corticosteroids, including growth retardation [1,3].
There are no adequate and well-controlled studies in pregnant women. Pregnant women were excluded from the clinical studies conducted with calcipotriol/betamethasone foam.
Calcipotriol/betamethasone foam should only be used during pregnancy if the potential benefit to the patient justifies the potential risk to the fetus.
Animal reproduction studies have not been conducted with calcipotriol/betamethasone foam. However, calcipotriol has been shown to be fetotoxic and betamethasone dipropionate has been shown to be teratogenic in animals when given systemically [1,3].
It is not known whether topically administered calcipotriol or corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk.
Because many drugs are excreted in human milk, caution should be exercised when calcipotriol/betamethasone foam is administered to a nursing woman.
The patient should be instructed not to use the foam on the breast when nursing [1,3].
No interaction studies have been performed [1].
Calcipotriol/betamethasone foam should not be used concomitantly with the following medications, as they may occasionally cause excessive blood calcium levels:
Adverse effects in < 1% of recipients associated with the use of calcipotriol/betamethasone foam in clinical trials included [6,7]:
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).