Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand, 2001. Updated by Giri Raj, Dermatologist, New Plymouth; Dr Amanda Oakley, June 2014; with minor update September 2024.
Introduction
Classification
Prevalence
Clinical features
Symptoms
Causes
Diagnosis
Treatment
Other treatment options
A congenital melanocytic naevus (American spelling nevus) is a proliferation of benign melanocytes that are present at birth or develop shortly after birth. This form of a congenital naevus is also known as a brown birthmark.
Similar melanocytic naevi, or moles that were not present at birth, are often called ‘congenital melanocytic naevus-like’ naevi, ‘congenital type’ naevi or ‘tardive’ naevi.
Naevi may also form from other skin cells (eg, vascular naevi, which are formed from blood vessels). Some of these are also congenital (present at birth) and are described on other pages of DermNet.
Congenital melanocytic naevi are usually classified by their size in an adult. There are several different classifications.
A modification of the above criteria is used in some centres in an effort to increase the accuracy of classification.
In 2013, a new categorisation of congenital melanocytic naevi using predicted adult size was proposed:
Congenital melanocytic naevi should be described according to their body site, colours, surface features and whether or not there is hypertrichosis (hairs).
Congenital melanocytic naevi usually grow proportionally with the child. As a rough guide, the likely adult size of a congenital naevus can be calculated as follows:
Some congenital naevi are given specific descriptive names. Some of these are listed here.
Satellite lesions
Tardive naevus
Garment naevus
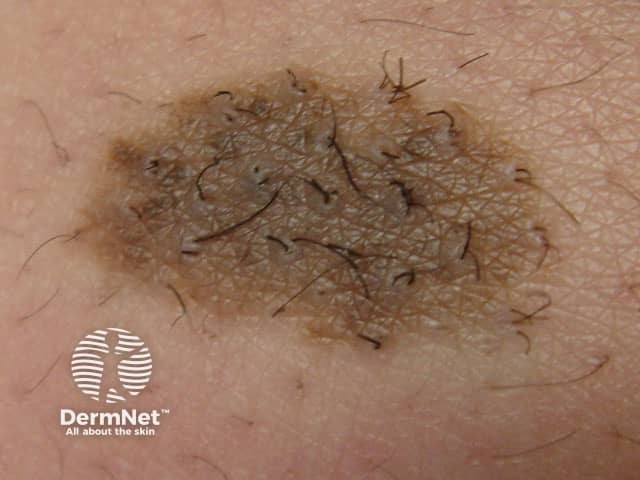
Congenital naevus
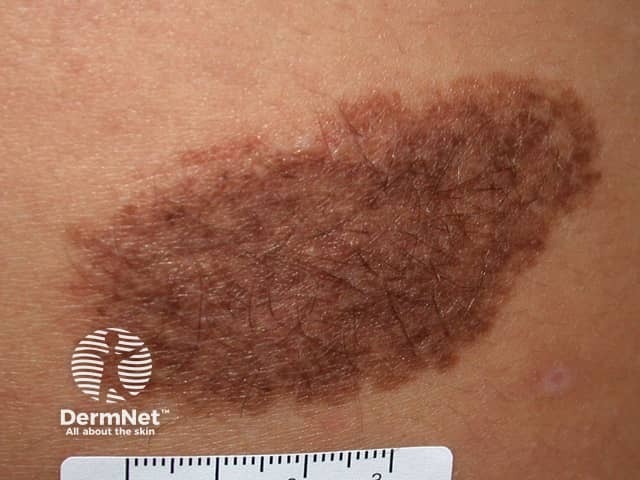
Congenital naevus
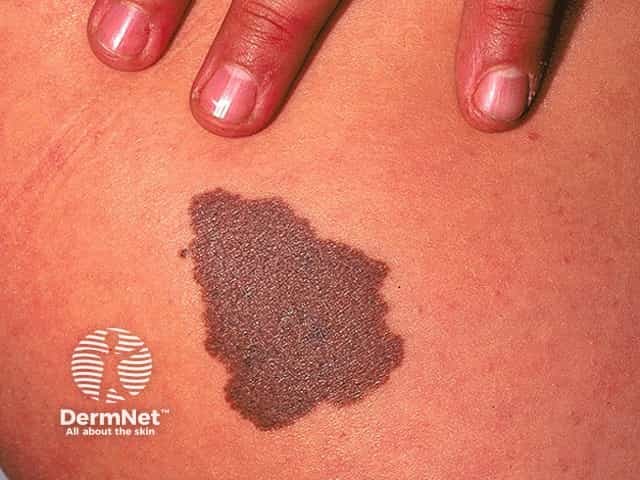
Congenital naevus
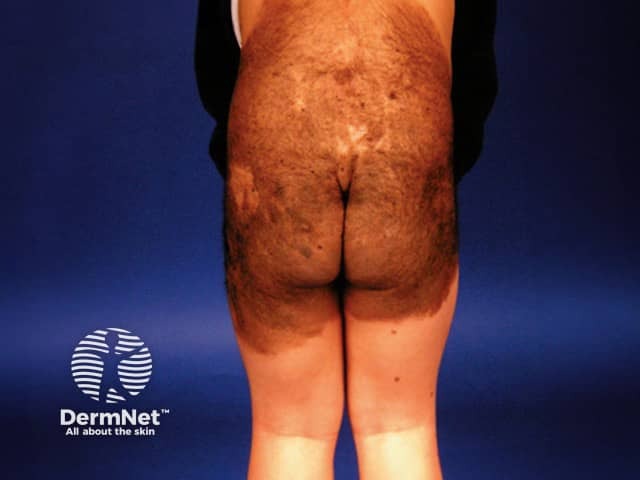
Congenital naevus
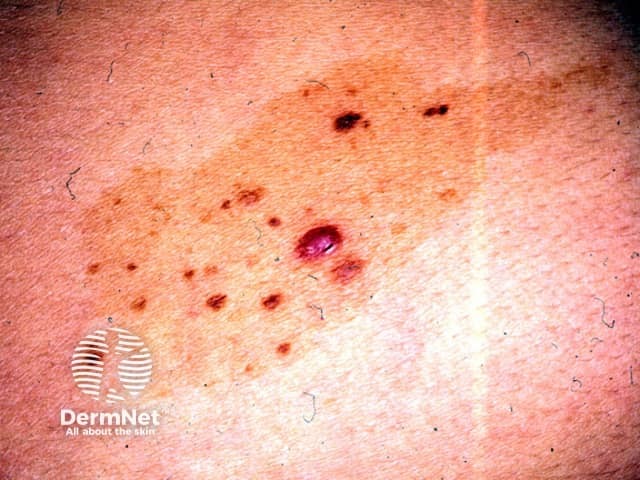
Congenital naevus
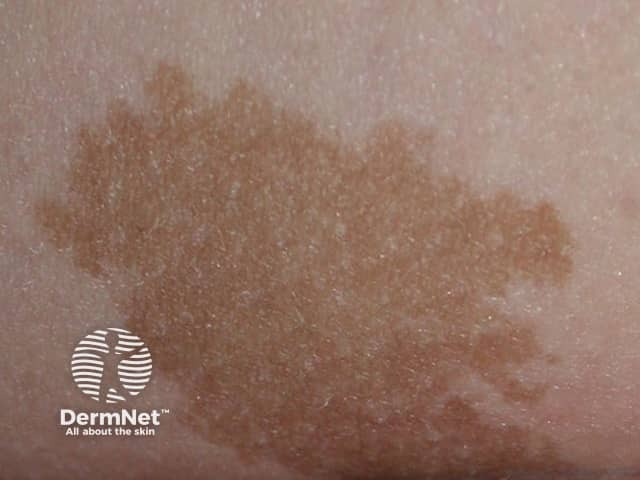
Congenital naevus
They occur in all races and ethnic groups, and males and females are at equal risk.
Congenital melanocytic naevi present as single or multi-shaded, round or oval-shaped pigmented patches. They may have increased hair growth (hypertrichosis). The surface may be slightly rough or bumpy.
Congenital naevi usually enlarge as the child grows but they may sometimes become smaller and less obvious with time. Rarely some may even disappear. However, they may also become darker, raised, more bumpy and hairy, particularly around the time of puberty.
Congenital melanocytic naevi are usually asymptomatic, however, some may be itchy, particularly larger lesions. It is thought there may be a reduced function of sebaceous (oil) and eccrine (sweat) glands, which may result in skin dryness and a heightened sensation of itch.
The overlying skin may become fragile and erode or ulcerate. Deep nests of melanocytes in the dermis may weaken the bonds between the epidermis and the dermis and account for skin fragility .
Congenital melanocytic naevi are often unsightly, especially when extensive, ie large or giant congenital melanocytic naevi. They may, therefore, result in anxiety and impaired self-image, especially when the lesions are in visible areas.
Giant melanocytic naevi, and to a lesser degree small lesions, are associated with increased risk of developing cutaneous melanoma, neurocutaneous melanoma and rarely other tumours (see below).
Congenital melanocytic naevi are caused by localised genetic abnormalities resulting in the proliferation of melanocytes; these are cells in the skin responsible for normal skin colour. This abnormal proliferation is thought to occur between the 5th and 24th weeks of gestation. If proliferation starts early in development, giant and medium-sized congenital melanocytic naevi are formed. Smaller congenital melanocytic naevi are formed later in development after the melanoblasts (immature melanocytes) have migrated from the neural crest to the skin.
In some cases, there is also overgrowth of hair-forming cells and epidermis, forming an organoid naevus.
Very early onset of congenital naevus before the separation of the upper and lower eyelids results in kissing naevi, ie one part of the naevus is on the upper lid and the other part is on the lower eyelid.
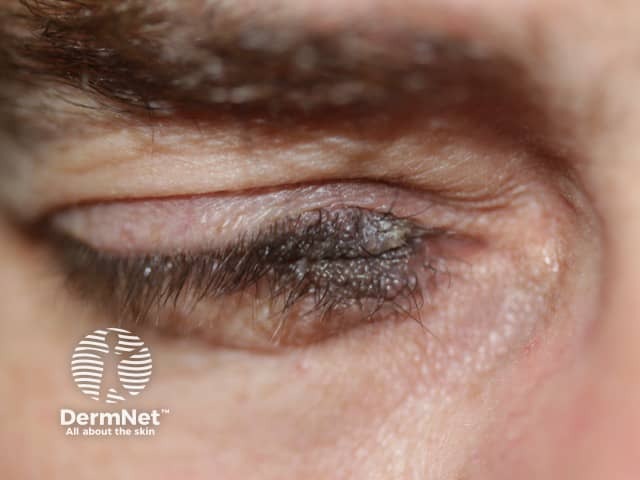
Kissing naevus
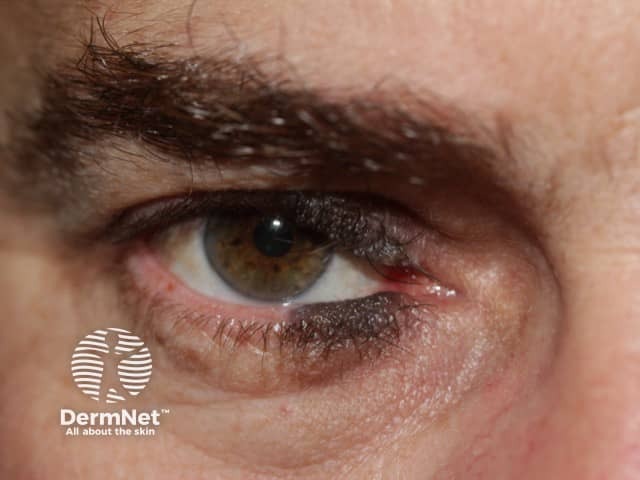
Kissing naevus
Proto-oncogenes c-met and c-kit have important roles in the development of melanocytes. Hepatocyte growth factor, a cytokine (messenger protein) that regulates the proliferation and migration of melanocytes, may also be important in the development of congenital melanocytic naevi.
Neurocutaneous melanosis is a rare syndrome defined by the proliferation of melanocytes in the central nervous system (brain and spinal cord) and the presence of a congenital melanocytic naevus. The majority of cases are associated with a giant congenital melanocytic naevus and satellite lesions.
The estimated risk of NCM developing in individuals with large and giant congenital melanocytic naevus (CMN) is 10–33%. However, it is likely that the majority of cases remain asymptomatic, and the true incidence remains unknown. The melanocytes in the brain and spinal cord may often be detected by an MRI scan.
Neurocutaneous melanocytosis may present with symptoms of raised intracranial pressure, such as:
The diagnosis of a congenital melanocytic naevus is usually based on the clinical appearance. If there is any doubt, examining the lesion with dermoscopy or taking a sample of the lesion for histology (biopsy) may show characteristic microscopic features.
Evaluation of the congenital melanocytic naevus by dermoscopy will reveal the pattern of pigmentation and its symmetry or lack of symmetry. The most common global pattern of congenital or tardive melanocytic naevus is globular, but reticular, structureless and mixed patterns may occur. The naevus may have differing structures across the lesion, sometimes leading to overall asymmetry of the structure.
Congenital melanocytic naevi are usually larger than acquired naevi (which are melanocytic naevi that appear after 2 years of age), and the naevus cells often extend deeper into the dermis, fat layer, and deeper structures. The naevus cells characteristically cluster around blood vessels, hair follicles, sebaceous and eccrine glands, and other skin structures. Congenital naevus cells tend to involve collagen bundles in the deeper layers of the skin more than is the case in an acquired naevus.
The following characteristics of congenital melanocytic naevus are associated with the increased risk of development of melanoma (a skin cancer).
The risk of melanoma is mainly related to the size of the congenital melanocytic naevus. Small and medium-sized congenital melanocytic naevi have a very small risk, well under 1%. Melanoma is more likely to develop in giant congenital naevi (lifetime estimates are 5–10% but may be overestimates). A review of 27 studies including over 11,000 patients with congenital naevi, the prevalence of melanoma was 1.84 overall, and 2.73% in those with large congenital naevi. Melanoma can start deep inside the naevus or within any neuromelanosis found in the brain and spinal cord. Very rarely, other tissues that contain melanocytes may also be a source of melanoma such as the gastrointestinal tract mucosa. In 24% of cases, the origin of the melanoma cannot be identified.
Melanoma associated with a giant congenital melanocytic naevus or neuromelanosis can be very difficult to detect and treat.
The risk of development of melanoma is greater in early childhood; 70% of melanomas associated with giant congenital melanocytic naevi are diagnosed by the age of ten years.
Rarely, other types of tumour may develop within giant congenital melanocytic naevi including benign tumours (lipomas, schwannomas) and other malignant tumours (including sarcomas).
Melanoma can also develop within a small congenital melanocytic naevus. This is rare and likely to occur on the periphery of the naevus during adult life.
Unfortunately, when a rare melanoma arises within a giant congenital melanocytic naevus, the prognosis is unfavourable. This is due to the deeper origin of the tumour rendering it more difficult to detect on clinical examination, resulting in a later stage at presentation. The deeper location also facilitates earlier spread through blood and lymph vessels. In 24% of cases, the melanoma has already spread to other sites (metastases) at the time of the first diagnosis.
Management of a congenital melanocytic naevus must take into account the age of the subject, the lesion size, the location and depth, and the risk of developing malignant change within the lesion.
The only definite indication for surgery in a giant congenital melanocytic naevus is when melanoma develops within it.
If a small congenital naevus is growing at the same rate as the child and is not changing in any other way, the usual practice is not to remove it until the child is old enough to co-operate with a local anaesthetic injection, usually around the age of 10 to 12 years. Even then, removal is not essential.
Reasons to consider surgical removal may include:
The following factors should be considered prior to prophylactic surgical removal of a naevus.
Complications that may occur after surgery include:
Dermabrasion can allow partial removal of a large congenital naevus; deeper naevus cells may persist. Dermabrasion may lighten the colour of the naevus but may not reduce hair growth within it. It can cause scarring.
Tangential or shave excision uses a blade to remove the top layers of the skin (epidermis and upper dermis). This may reduce the pigmentation but the lesion may not be completely removed. Shave excision may result in significant scarring.
Chemical peels using trichloroacetic acid or phenol may lighten the pigmentation of a superficial (surface) congenital naevus that is located in the upper layers of the skin.
Laser treatment is considered if surgical intervention is not possible. They may result in lightening of the lesion. Suitable devices include:
Techniques that result in partial removal of a congenital naevus can make the lesion more difficult to assess during long-term surveillance.