Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Lesions (cancerous)
Author: Matthew Howard, Clinic/Research Fellow, Victorian Melanoma Service, Melbourne, VIC, Australia. DermNet Editor in Chief: Adjunct A/Prof. Amanda Oakley, Dermatologist, Hamilton, New Zealand. Copy edited by Gus Mitchell. November 2019.
Introduction
Demographics
Introduction
Contraindications
Introduction - neoadjuvant therapy
Benefits
Disadvantages
Side effects and risks
Adjuvant therapy is defined as a treatment given after the initial therapy. In the case of cutaneous melanoma, adjuvant therapy is a form of treatment considered after complete surgical excision of the melanoma (the first-line treatment) [1].
Adjuvant immunotherapy aims to eliminate residual microscopic melanoma cells via the immune system, to reduce the risk of future cancer recurrence, and to improve the overall chance of cure [1].
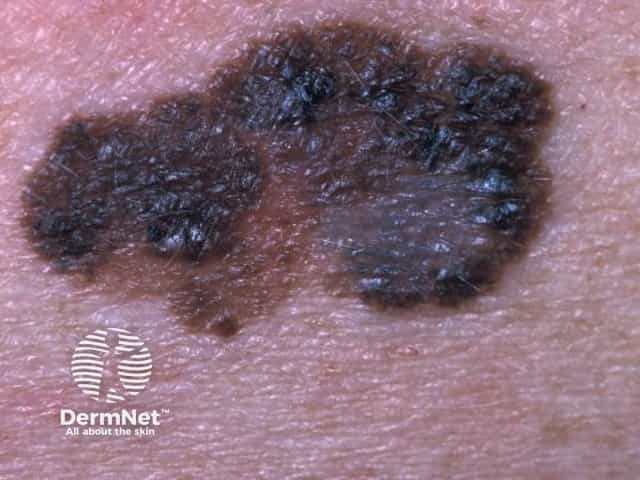
Superficial spreading melanoma
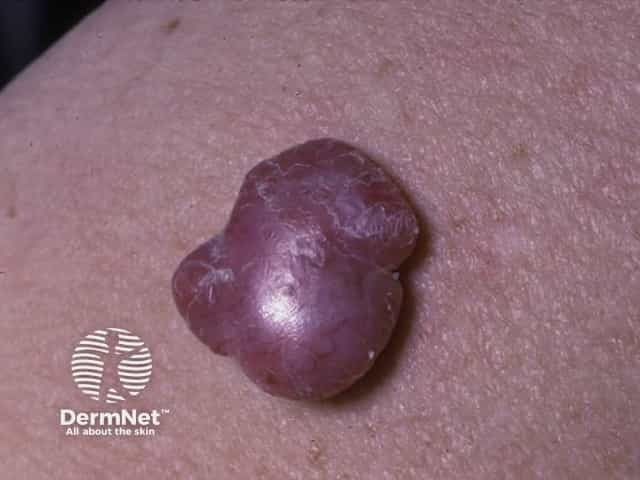
Nodular melanoma
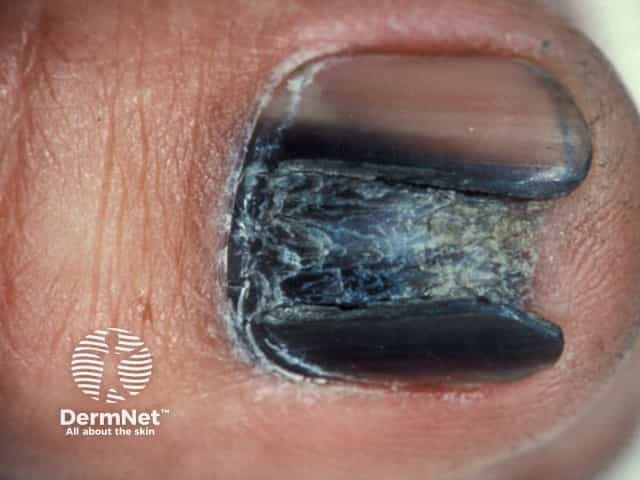
Acral melanoma
Indications for consideration of adjuvant therapies for cutaneous melanoma include [1–3]:
Immunotherapy and targeted cancer therapy against driver mutations have proven a benefit in stage IV metastatic melanoma (when distant metastases have been detected). Adjuvant therapy is also reported to reduce the relative risk of recurrence by 40% in stage III metastatic melanoma with a 15–25% absolute reduction in local and distant recurrence risk. Overall survival data are not yet reported for immunotherapy, but significant overall survival benefits are seen with adjuvant-combined dabrafenib and trametinib in BRAF–mutant stage III metastatic melanoma.
The results of adjuvant treatment have been published using immunotherapy, targeted therapy, or radiotherapy that has been given for up to a total of 12 months.
There are two classes of immune checkpoint inhibitors, as of 2019.
Immunotherapies are given by intravenous infusion every three weeks [4].
Targeted therapies block the mitogen-activated protein (MAP) kinase pathway [6].
BRAF inhibitors are administered orally twice daily; a MEK inhibitor is usually taken orally once daily [8]. A BRAF and MEK inhibitor are usually used together in combination therapy to limit early tumour resistance to single-agent therapy [7].
Stereotactic external beam radiotherapy can be considered after therapeutic lymphadenectomy to reduce the rate of nodal recurrence, particularly when there is an extranodal extension of metastatic melanoma [8,9].
Typically, patients with lower risk or thin localised melanoma are not currently offered adjuvant therapies (Stage I/IIA disease) [1,2].
The relative contraindications for the use of immunotherapy include:
Targeted therapy using BRAF inhibitors is contraindicated for patients without the BRAF V600 driver mutation [7].
Unlike adjuvant therapy, which is given after surgery to remove melanoma, neoadjuvant therapy is an induction treatment commenced before the excision. It is typically used when the primary melanoma is unresectable (ie, it is a large tumour, the excision defect can’t be closed, or the excision would be unacceptable cosmetically) or when any nodal or visceral melanoma metastases are too bulky to safely remove. The intention of neoadjuvant therapy is to reduce the size of the tumour to allow the surgery to take place [14].
Early results of clinical research programmes are demonstrating consistent, clinically meaningful benefits from targeted therapies and immunotherapies in stage III metastatic melanoma when the melanoma has been completely resected.
Initial research data on immunotherapy using ipilimumab in stage III metastatic melanoma is from two major studies. The chance of overall survival at 5 years was improved by 28%, and the risk of metastatic melanoma recurrence at 5 years was improved by 24% in those randomised to ipilimumab compared to placebo [15].
Studies into BRAF inhibitors and BRAF/MEK dual inhibition for patients with stage III metastatic melanoma were conducted prior to the immunotherapy studies. Key results are listed below. A consistent improvement in the overall survival (OS) of patients was demonstrated with the use of single agent MEK/BRAF inhibition compared to placebo. This benefit was increased with dual inhibition [7,16]. Due to the adverse effects of targeted therapy, one of the components may need to be stopped [17].
Inhibitor |
CR + PR% |
Median PFS, Median OS |
|---|---|---|
Vemurafenib [18] |
53% |
6.9 months, 13.6 months |
Dabrafenib [19] |
50% |
6.7 months, 18 months |
Trametinib [20] |
22% |
4.8 months (PFS only) |
Dabrafenib + trametinib [7] |
67% |
11 months, 25.1 months |
Dabrafenib + trametinib versus vemurafenib [16] |
64% |
12.6 months, 25.6 months |
CR: complete response; PR: partial response; CR + PR%: percentage of patients with a partial or complete response; PFS: progression-free survival; OS: overall survival
Local control of melanoma may be enhanced by adjuvant radiotherapy if the risk of local recurrence remains unacceptably high after surgical excision such as with:
Although randomised controlled trials have not evaluated the benefit of adjuvant radiotherapy, several large observational cohorts have reported positive results with 50–85% improvement in risk of local recurrence. Possible evidence was also observed for decreased nodal recurrence [22–24].
The local inflammatory effects resulting from radiation therapy may provide a synergistic boost to the efficacy of immunotherapy [25,26]. Further research is underway to evaluate its benefits [27,28].
The comparative benefits of immunotherapy to traditional chemotherapy have been game-changing for the treatment of metastatic melanoma in some patients. However, in many patients, melanoma does not respond to immunotherapy or only partially responds. In addition, targeted therapies are only indicated for those whose melanoma carries the BRAF V600 driver mutation (approximately 40–50% of cutaneous melanoma) [29].
There are questions still to be answered about adjuvant therapy for melanoma.
Adjuvant treatments are generally well tolerated with comparative greater short-term toxicity with BRAF/MEK combined inhibition compared to immunotherapy; however, there is a risk of permanent toxicity with adjuvant immunotherapy.
Immunotherapy can result in side effects due to immune activation [13,30]. The rates described below are for PD1 inhibitors (nivolumab and pembrolizumab); immune-related adverse events are typically more frequent with CTLA-4 inhibition (ipilimumab).
See also Cutaneous adverse effects of checkpoint inhibitors.
Combined dabrafenib/trametinib causes the following adverse effects [17].
Acute side effects of radiotherapy may include [31,32]:
Late side effects of radiotherapy may include:
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).