Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Dr Maneka Gnanasegaram, Dermatology Registrar, Waikato Hospital, Hamilton, New Zealand, 2011.
Introduction
Demographics
Clinical features
Diagnosis
Treatment
Outlook
Melanoma is a type of skin cancer that develops from the pigment-producing cells (melanocytes) of the skin, mucosa, eye, and rarely other sites. Metastatic melanoma is melanoma that has spread to other sites of the body. The spread occurs through the lymphatic system and/or the blood vessels. Melanoma can spread to the subcutaneous tissue which lies underneath the skin, the lymph nodes, and to other organs such as the lungs, liver, bone or brain.
Metastatic melanoma can be classified into local recurrence, in transit metastasis, nodal metastasis, and haematogenous spread.
Local recurrence is defined as a recurrence of melanoma within 2cm of the surgical scar of primary melanoma. It can result either from direct extension of the primary melanoma or from spread via the lymphatic vessels.
In-transit metastases are melanoma deposits within the lymphatic vessels more than 2 cm from the site of the primary melanoma.
Nodal metastasis is metastatic melanoma involving the lymph nodes. Every site on the body drains initially to one or two nearby lymph node basins. The lymph nodes first involved are the regional lymph nodes. Usually, the involved lymph nodes become enlarged and may be be felt as a lump through the skin.
Haematogenous spread is spread of melanoma cells in the bloodstream, which can happen either by a tumour invading blood vessels or secondary to lymph node involvement. Once in the bloodstream, melanoma cells can travel to distant sites in the body and deposit. It can proliferate in any tissue but most often grows in the lungs, in or under the skin, the liver and brain. Many patients also develop metastases in bone, gastrointestinal tract, heart, pancreas, adrenal glands, kidneys, spleen and thyroid.
Diffuse melanosis cutis is a rare presentation of metastatic melanoma in which the entire skin surface changes colour.
Melanoma-associated leukoderma is depigmentation that occurs in patients with metastatic melanoma. It may present as white areas within a melanoma due to regression, halo melanoma, or as white patches resembling vitiligo distant from the melanoma. Leukoderma associated with metastatic melanoma is also known as melanoma-associated vitiligo.
Leukoderma can also be triggered by drugs used to treat metastatic melanoma, such as ipilimumab, immune checkpoint inhibitors (pembrolizumab, nivolumab), and BRAF inhibitors (vemurafenib, dabrafenib) [see also Drug-induced vitiligo].
Melanoma usually starts as a single lesion on the skin or mucous membrane. This lesion can progress to the formation of metastases if it is not recognised and treated effectively at an early stage.
Risk factors for the development of melanoma include:
The risk of melanoma metastasising is highest in an individual with an aggressive rapidly growing melanoma, an unrecognised melanoma, advanced primary melanoma, melanoma that was not completely excised (or not removed at all), and/or is immunosuppressed.
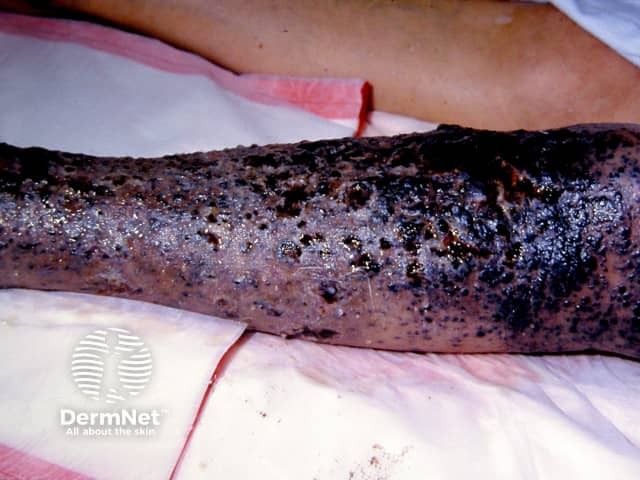
Cutaneous melanoma metastases usually grow rapidly within the skin (cutaneous metastases) or under the skin surface (subcutaneous metastases); dermal metastases are more common than subcutaneous. They are usually firm or hard in consistency. Cutaneous metastases may be any colour but are often black or red. They may also ulcerate and bleed.
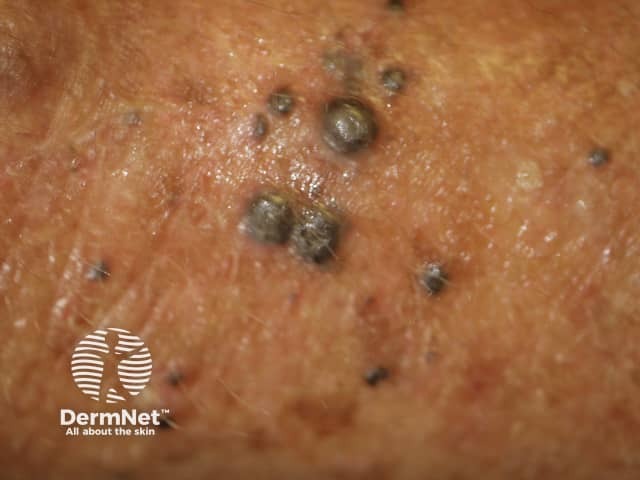
Metastatic melanoma
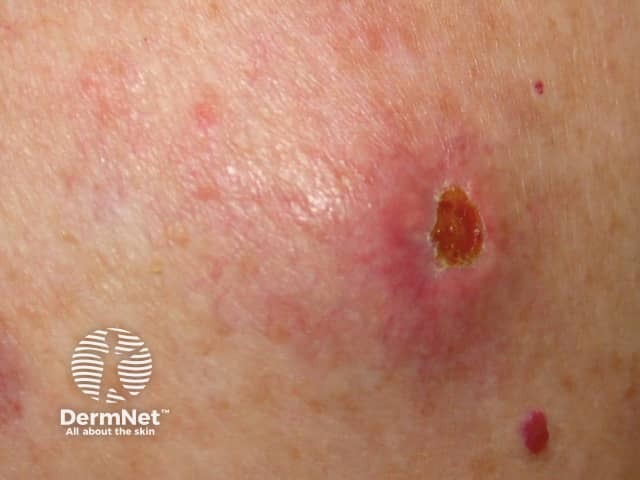
Metastatic melanoma
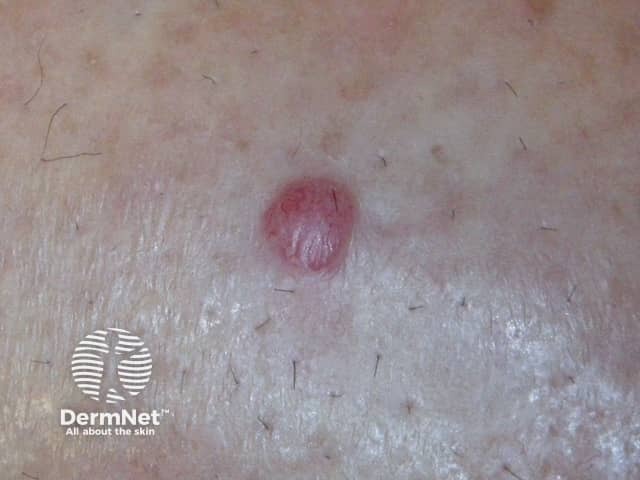
Metastatic melanoma
Epidermotropic metastatic melanoma is rare. In this case, the metastases develop more superficially than usual, within the epidermis. Epidermotropic metastatic melanoma is often initially misdiagnosed as the primary melanoma. The diagnosis of epidermotropic metastatic melanoma should be considered if multiple lesions arise with similar pathology.
Subcutaneous metastases are skin coloured or bluish lumps. They are usually painless.
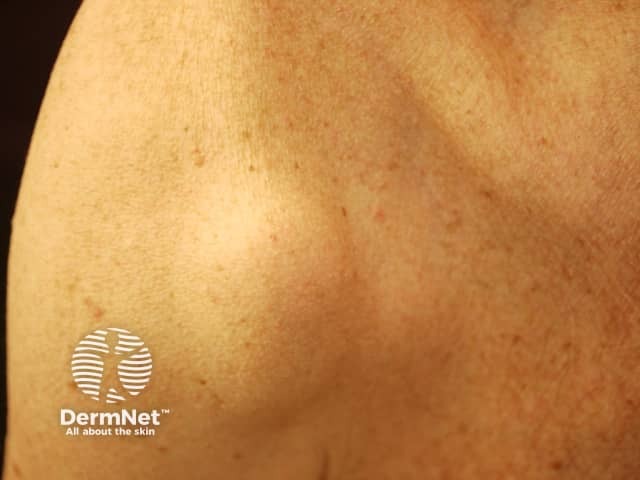
Metastatic melanoma
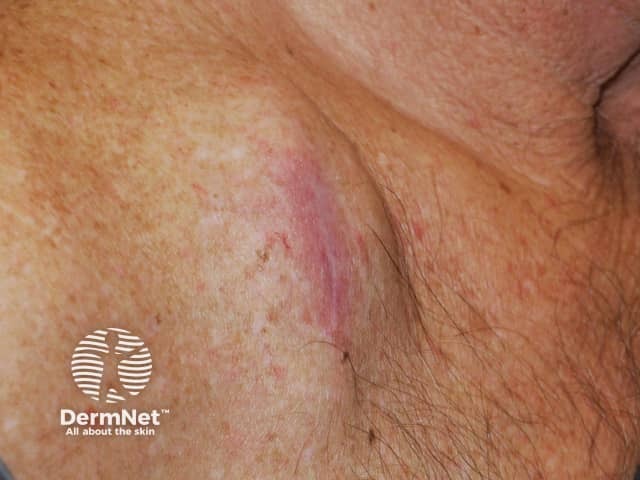
Metastatic melanoma
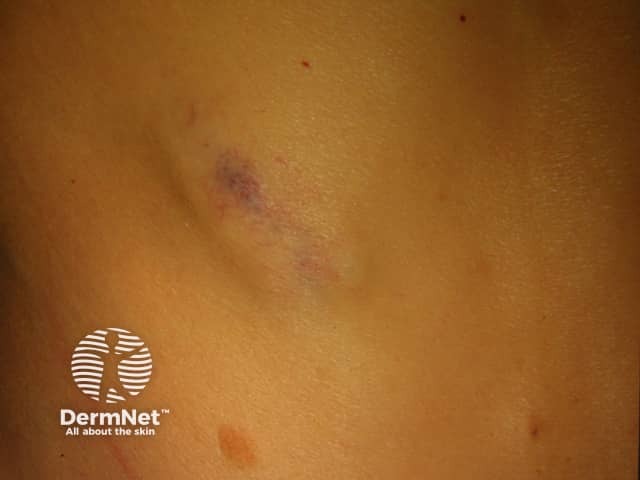
Metastatic melanoma
Obstruction of lymphatic vessels due to melanoma in the lymph nodes or surgical removal of the lymph glands can result in swelling of the associated limb (lymphoedema).
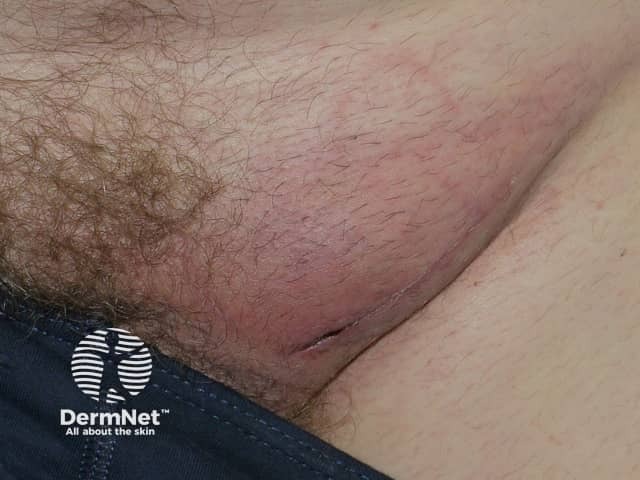
Nodal melanoma
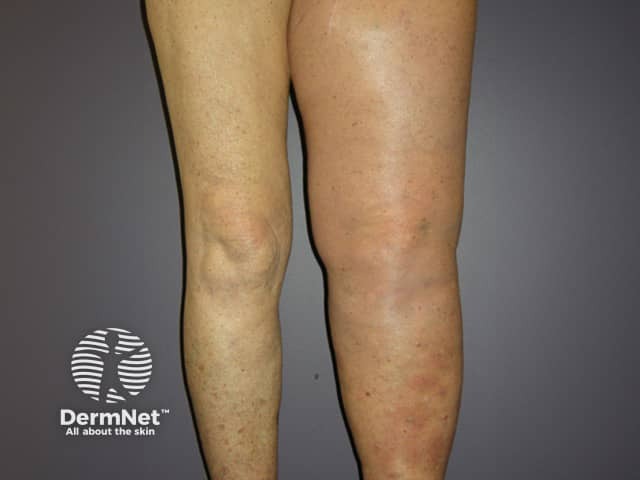
Swollen leg due to nodal melanoma
In transit metastases from primary melanoma on foot
See more images of metastatic melanoma.
Metastatic melanoma may be diagnosed on the basis of characteristic skin lesions.
Other clues are enlarged lymph nodes and/or symptoms related to other organs. People with metastatic melanoma may feel tired, lethargic, and have weight loss prior to the diagnosis being made.
Histopathology
Blood tests
In some cases, a deposit of metastatic melanoma can be surgically removed. This is an appropriate treatment option with the aim of cure when there is a solitary metastasis of the skin, in the subcutaneous tissue or of a lymph node. Sometimes surgery is done when there is felt to be one metastasis to another organ such as the liver. Surgery may also be helpful in reducing symptoms and improving quality of life, even if it will not be expected to cure the disease.
Chemotherapy can slow the rate of progression of metastatic melanoma but it does not cure it. Agents such as dacarbazine, temozolomide and combinations of other agents are used. A chemotherapy agent is sometimes successfully infused into a limb to treat in-transit metastases (isolated limb perfusion) but this technique can have serious complications.
Electrochemotherapy is a new treatment that uses electrical impulses to enhance effectiveness bleomycin or cisplatin injected directly into skin metastases.
High dose interferon-α is proven to improve survival and lower relapse rate in high-risk melanoma. Side effects can be difficult to tolerate for many people and can even be fatal. They include flu-like symptoms, tiredness, depression and rhabdomyolysis (destruction of muscle tissue). See Adjuvant therapies for melanoma.
Radiotherapy can be useful for improving symptoms caused by metastatic disease and along with surgery is the mainstay of treatment of brain metastases.
Immunotherapy for melanoma includes interleukin-2, which is given intravenously. Side effects include fever, low blood pressure and irregular heart rhythms.
Interferon alfa 2b is delivered by subcutaneous or intravenous injection. Side effects include 'flu-like symptoms, depression, and depression cell counts.
Topical imiquimod is an immune modifier that has been used alone or in combination with intravenous interleukin-2 treatment. Imiquimod cream is applied directly to melanoma metastases to enhance an immune response to the tumour. The encouraging response of in-transit metastases has been reported in some cases.
Topical diphenylcyclopropenone or diphencyprone in various concentrations (0.0001% to 10%) in solution or cream may be useful for small cutaneous melanoma metastases. The first application sensitises the patient to the chemical over about 10 days. Further applications applied to the lesions at weekly intervals cause allergic contact dermatitis, which can be very itchy and uncomfortable and may generalise. When effective, existing treated lesions stop enlarging and may shrink or disappear. Dramatic responses have been reported including regression of involved lymph nodes.
Intralesional immunotherapy for melanoma metastases using talimogene laherparepvec (T-VEC), Allovectin-7® and Rose Bengal is under investigation.
With a range of new therapies being developed and studied for melanoma, some patients choose to participate in a clinical trial. This can mean having access to a treatment that wouldn’t otherwise be possible.
A 2018 Cochrane report confirmed that immune checkpoint inhibitors and small-molecule targeted drugs have significantly improved patient prognosis, especially combinations of immune checkpoint inhibitors and combinations of small-molecule targeted drugs, but toxicity is common.
A small, preliminary study suggested that beta-blockers may prevent or delay the recurrence of melanoma.
Palliative care focuses on quality of life rather than the length of life. It includes control of pain and other uncomfortable symptoms and provides support for patients and their families.
In most instances, it is not possible to cure metastatic melanoma entirely because it tends to spread to multiple sites. Treatment is focussed on improving the quality of life and the length of survival.
The prognosis of melanoma depends on the disease staging, which is based around characteristics of the primary tumour, nodal and distant metastases. The prognosis is poorer with higher numbers of involved nodes and with metastases to internal organs and distant sites.