Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Lesions (cancerous)
Author: Anoma Ranaweera, Medical Writer, Auckland, New Zealand. DermNet Editor in Chief: Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand, January 2015. Updated August 2018. Copy edited by Gus Mitchell.
Introduction
Uses
How it works
Administration
Potential drug interactions
Adverse effects
Cutaneous adverse effects
Reporting
Use in pregnancy
Use of nivolumab
Paediatric use
Geriatric use
Renal impairment
Hepatic impairment
Nivolumab (OPDIVO®, Bristol Myers Squibb, New Jersey, USA) is a human programmed death receptor-1 (PD-1)-blocking antibody, approved for treatment of advanced melanoma.
In December 2014, the US Food and Drug Administration (FDA) approved the use of nivolumab in:
In October 2015, nivolumab was approved for use in combination with ipilimumab for improved response in advanced melanoma (unresectable or metastatic disease). In January 2016, it was also approved in untreated BRAF-wild type melanoma. In December 2017, the FDA approved nivolumab for the adjuvant treatment of patients with melanoma and involvement of lymph nodes or metastatic disease who have undergone complete resection.
In the European Union, the European Medicines Agency (EMA) has validated for review the Marketing Authorization Application for Nivolumab in advanced melanoma. The application has also been granted accelerated assessment by the EMA’s CHMP (Committee for Medicinal Products for Human Use). Nivolumab is available in New Zealand, and funded by PHARMAC for some cases of metastatic and unresectable advanced melanoma.
In June 2014, the European Commission approved nivolumab a PD-1 immune checkpoint inhibitor, for the treatment of advanced (unresectable or metastatic) melanoma in adults, regardless of BRAF status. The approval allows for the marketing of nivolumab in all 28 Member States of the EU. It follows an accelerated assessment by the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP), which was announced on 24 April, 2015. Approval was based on CheckMate-066 trial demonstrating superior overall survival vs. dacarbazine in the first-line setting and CheckMate-037 trial showing improved response vs. chemotherapy in previously treated patients.
In July 2018, the European Commission (EC) approved nivolumab for the adjuvant treatment of adult patients with melanoma with involvement of lymph nodes or metastatic disease who have undergone complete resection. This indication is for both BRAF mutant and wild-type melanoma patients. With this decision, nivolumab becomes the first and only PD-1 therapy to receive an EC approval in the adjuvant setting. The approval was based on results from the ongoing phase 3 randomized, double-blind CheckMate-238 trial.
Nivolumab is available in New Zealand, and funded by PHARMAC for some cases of metastatic and unresectable advanced melanoma.
Link to key clinical-trial evidence about nivolumab
In clinical trials the following adverse events have been observed at an incidence > 10%: increased alanine aminotransferase (28%), hyponatraemia (25%), increased alkaline phosphatase (22%), rash (21%), pruritus (19%), cough (17%), increased ALT (16%), hyperkalaemia (15%), upper respiratory tract infection (11%).
Other clinically important adverse events occurring at an incidence of 1–10% have included:
A clinical trial of the combination of nivolumab and ipilimumab had 36% dropout due to side effects, mainly diarrhoea and elevated liver function tests.
Skin complications are common in patients treated with nivolumab, with about 40% of patients experiencing these, with mean onset of rash being about 10 months into the course of treatment. Reported reactions include:
Darkening of hair has been reported in a few patients taking PD-1 inhibitors for non-small cell lung cancer [1].
See also Cutaneous adverse effects of checkpoint inhibitors.
For continued monitoring of the benefit and risk balance of nivolumab, healthcare professionals are asked to report any suspected adverse reactions to the New Zealand Pharmacovigilance Centre.
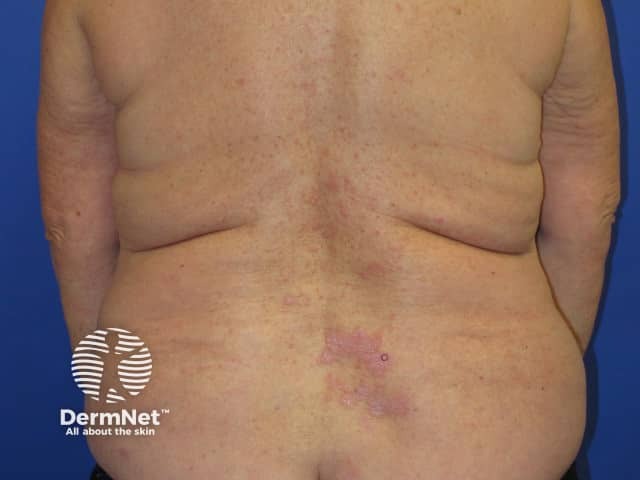
Nivolumab-induced lichenoid dermatitis
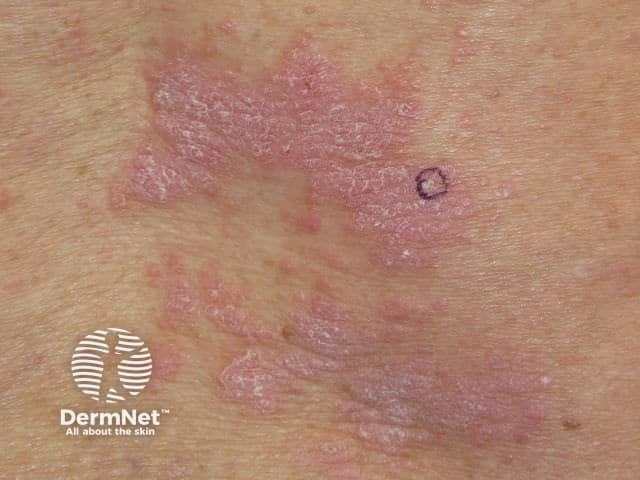
Nivolumab-induced lichenoid dermatitis
The safety and effectiveness of nivolumab has not been established in paediatric patients. Nivolumab should not be used in children below 18 years of age.
Clinical studies of nivolumab did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger patients. No overall differences in safety or efficacy were reported between elderly (≥ 65 years) and younger patients (< 65 years). No dose adjustment is required for elderly patients (≥ 65 years).
Based on a population pharmacokinetic analysis, no dose adjustment is recommended in patients with renal impairment.
Based on a population pharmacokinetic analysis, no dose adjustment is recommended for patients with mild hepatic impairment. Nivolumab has not been studied in patients with moderate or severe hepatic impairment.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).