Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Authors: Dr Amanda Oakley, Dermatologist, New Zealand, 2001. Updated: Dr Annabelle Yi Zhang, Auckland City Hospital, New Zealand; Honorary Associate Professor Paul Jarrett, Dermatologist, Middlemore Hospital and Department of Medicine, The University of Auckland, New Zealand. Copy edited by Gus Mitchell. January 2021.
Introduction
Demographics
More information
Uses
Side effects and risks
Contraindications
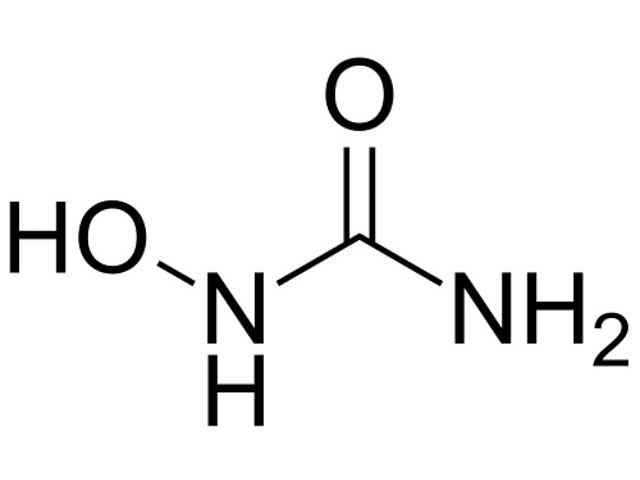
Hydroxyurea (CH4N2O2), also known as hydroxycarbamide, is an enzyme inhibitor used mainly to treat blood disorders. Hydroxyurea is usually administered as an oral capsule or tablet.
Hydroxyurea is frequently used in the treatment of chronic myeloproliferative diseases such as chronic myelocytic leukaemia, essential thrombocythaemia, and polycythaemia vera. It is also increasingly prescribed for sickle cell anaemia, and to treat refractory ovarian cancer and some head and neck cancers.
In dermatology, hydroxyurea has a role in treating psoriasis, and hypereosinophilic syndrome, and should be considered for human immunodeficiency virus-associated psoriasis.
Hydroxyurea was found to be an effective systemic treatment for psoriasis in 1970, but is used ‘off-label’. Psoriasis is not an approved treatment for psoriasis, mainly due to the availability of newer and/or more effective systemic treatments.
Hydroxyurea has been used off-label for the treatment of hypereosinophilic syndrome since 1978, usually in combination with systemic corticosteroids.
Hydroxyurea should not be used in pregnancy as it is teratogenic, and women of child-bearing age must be using reliable birth-control measures during hydroxyurea treatment and for at least six months after stopping. Men taking hydroxyurea should also use effective birth-control measures during treatment and for at least 12 months after completing treatment.
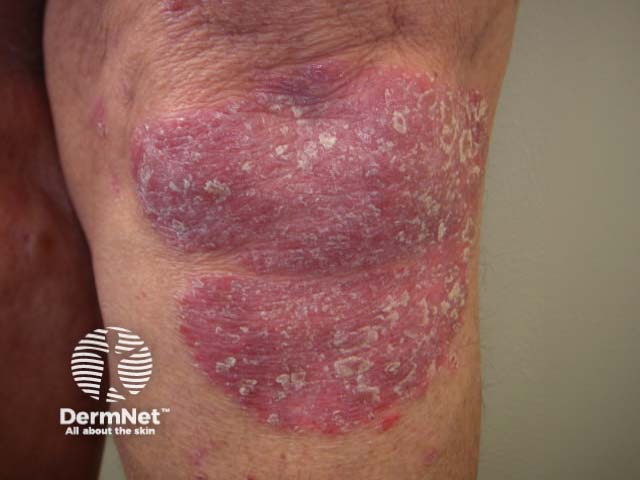
Chronic plaque psoriasis
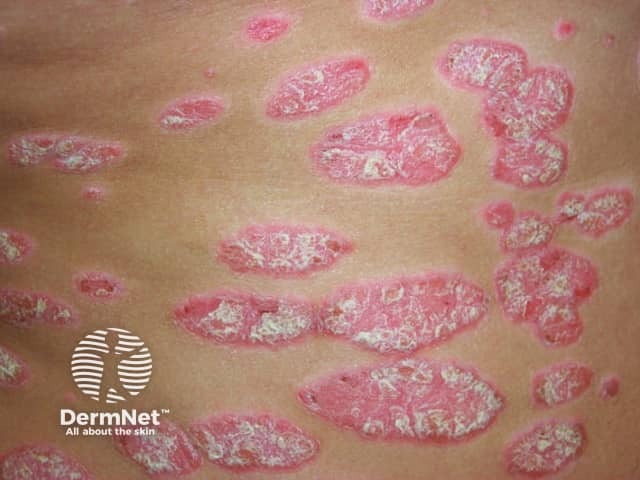
Chronic plaque psoriasis
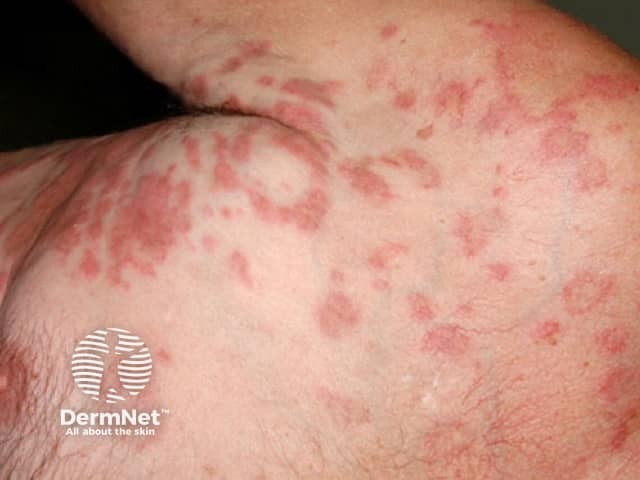
Hypereosinophilic syndrome
Hydroxyurea was first synthesised in the 1860s, but its use as a cancer therapy was not reported until 1960. Hydroxyurea inactivates the enzyme ribonucleotide reductase, therefore preventing DNA synthesis and repair. It synchronises the cell cycle in the G1 phase, and kills cells in the S phase.
The mechanism of action of hydroxyurea in treating psoriasis is assumed to be the inhibition of skin cell proliferation through its effects on DNA synthesis, and/or by causing gene hypomethylation which induces cell differentiation.
Enzyme inhibition by hydroxyurea also results in anti-retroviral effects, so could be considered in patients with HIV-associated psoriasis.
The oral bioavailability of hydroxyurea is almost 100% and it is distributed across all body tissues. The serum concentration peaks two hours after oral ingestion and has been cleared by 24 hours. Tissue concentration peaks at 8 hours and persists for 20 hours. Hydroxyurea is excreted by the kidneys.
Skin side effects are very common with hydroxyurea, especially with long-term or high-dose use, with studies reporting skin changes in up to 96% of patients when assessed by a dermatologist.
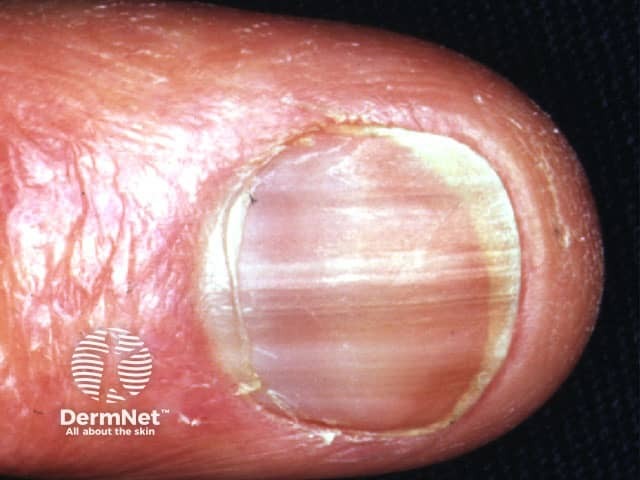
Nail pigmentation due to hydroxyurea
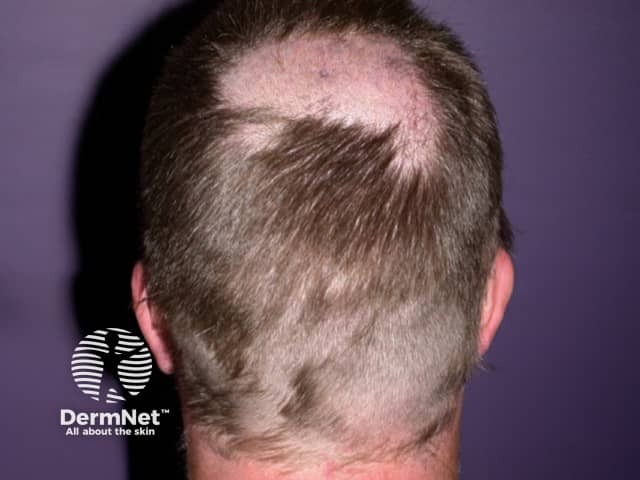
Drug-induced anagen effluvium
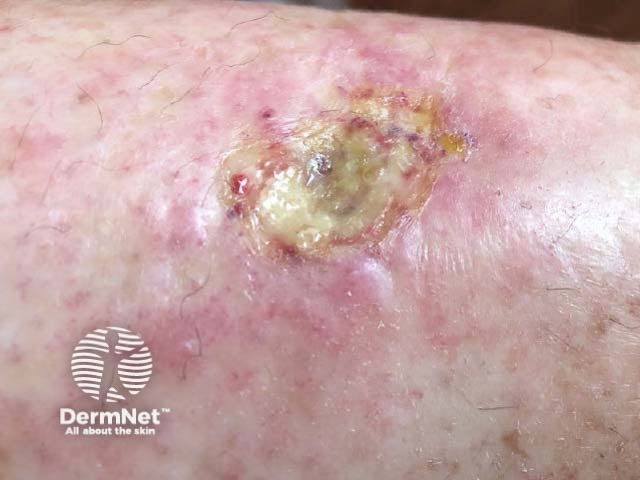
Hydroxyurea-induced leg ulcer
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).