Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Follicular disorder
Author: Dr Tim Aung, Primary Care Practitioner, Brisbane & Logan, Queensland, Australia. DermNet Editor in Chief: Adjunct A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand. June 2020.
Introduction
How they work
Indications
Adverse effects
Uses
Interactions with other drugs
Monitoring
Retinoids are a group of medications related to vitamin A. They are used to treat various inflammatory skin disorders, skin cancer, and skin ageing.
Retinoids are classified into four generations [1,2,3]:
Oral retinoids include:
The effects of retinoids are mediated through binding to and activation of the retinoic acid receptor (RAR) and the retinoid X receptor (RXR), which regulate cell proliferation, differentiation and apoptosis [4,5]. They also have a role in immune modulation, are anti-inflammatory, and activate tumour suppressor genes.
The therapeutic use of retinoids depends on the individual product [6,7]. Their specific usage varies at the approval of individual country and on the results of off-label trials [1,4,8,9,10].
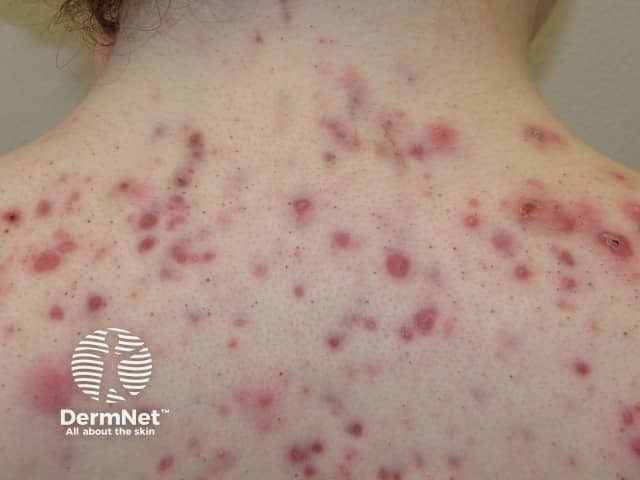
Severe acne
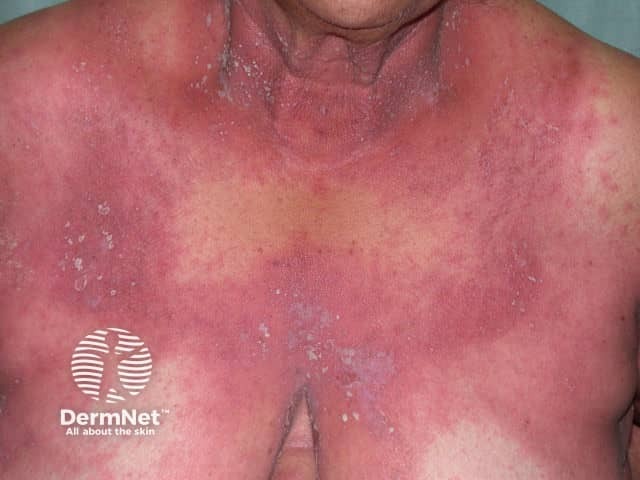
Generalised pustular psoriasis
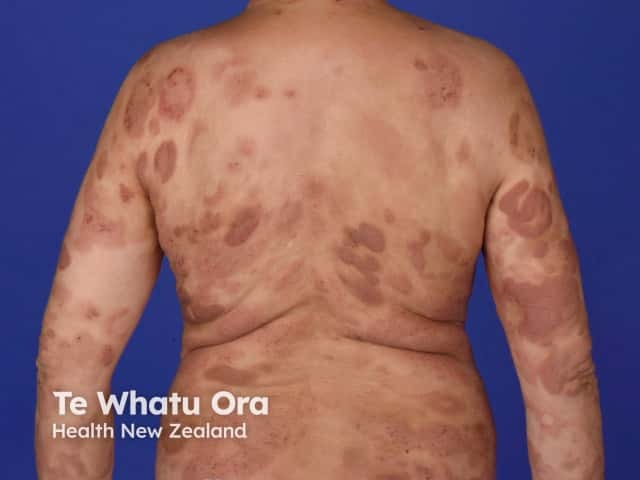
Cutaneous T-cell lymphoma
The most serious adverse effect of oral retinoids is teratogenicity (category X) if a retinoid is taken during pregnancy. The most frequent birth malformations caused by oral retinoids are craniofacial, central nervous system, cardiovascular, and thymic [11].
Systemic treatment may also cause:
Follow the instructions carefully.
Drug interactions with oral retinoids are uncommon and generally mild.
The effects of the oral retinoid and progress of the disease should be monitored at regular follow-up appointments.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).