Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author(s): Lauren Smith, Medical Student, University of Otago Christchurch, Christchurch, and Dr Adrian Sykes, Dermatology Registrar, Waikato Hospital, Hamilton, New Zealand. DermNet New Zealand Editor in Chief: Hon A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand. Copy edited by Maria McGivern. July 2017.
A systemic corticosteroid (or ‘steroid’) is a drug derived from cortisol, a natural steroid produced by the adrenal gland in the body. Systemic steroids are prescribed in dermatology for inflammatory skin diseases, such as:
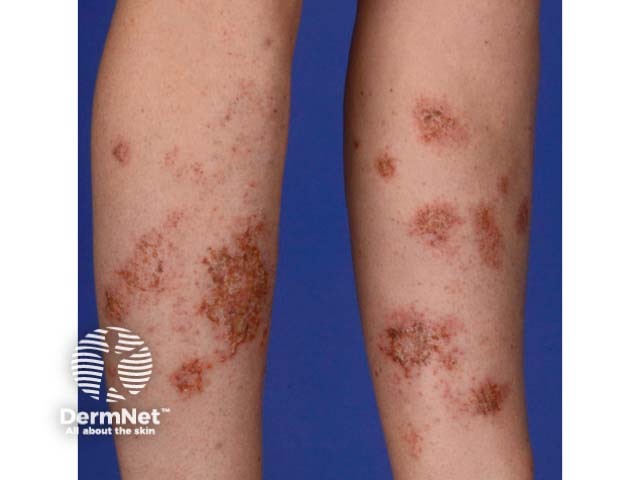
Discoid eczema
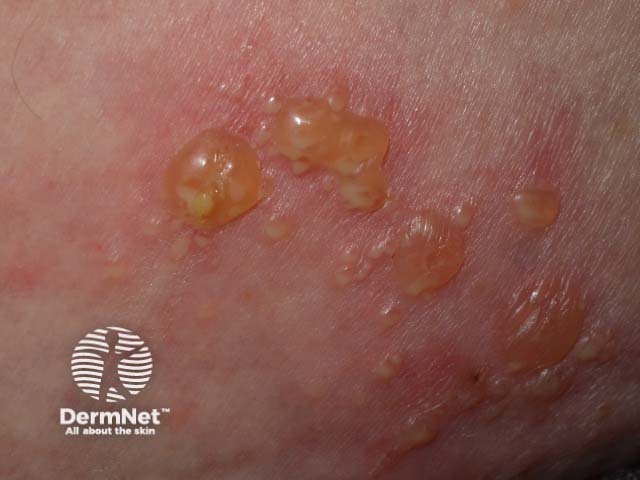
Bullous pemphigoid
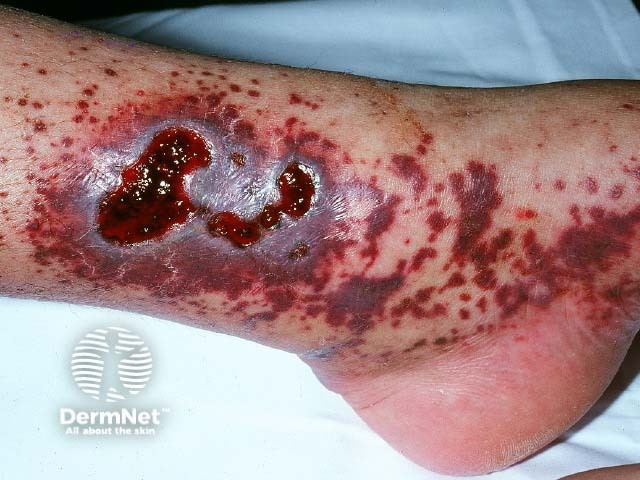
Cutaneous vasculitis
There are risks from the long-term effects of treatment with systemic steroids, including infections (bacterial, fungal and viral), hypertension, diabetes, osteoporosis, avascular necrosis, myopathy, and glaucoma.
There are several ways some of these risk factors can be reduced, including through continual assessment and monitoring, lifestyle changes, and prophylactic treatments.
The risks detailed below refer to patients treated with > 7.5 mg/day of prednisone (or equivalent doses of other corticosteroids) for periods of time greater than 3 months. Below these levels, the risk of health effects is negligible.
Systemic steroids stimulate bone resorption (breakdown) and reduce bone formation within the first few months of initiating treatment, which increases the risk of osteoporosis and fracture [1,2].
An assessment of bone mineral density (BMD) at the lumbar spine and/or the hip should be taken at the initiation of systemic steroids.
Consider a falls assessment for those individuals at increased risk of falls.
Blood tests are not usually necessary, but you could assess:
The risk of developing osteoporosis can be reduced through the implementation of lifestyle changes. These include:
The prevention of osteoporosis is controversial. A bisphosphonate (eg, alendronic acid, risedronate sodium, or zoledronic acid) is first-line therapy if the BMD-measured T-score is < −1 (ie, showing osteopenia or low bone mass) [4,5]. Osteoporosis is a T-score of < −2.5.
Bisphosphonates are also recommended for patients taking ≥ 7.5 mg/day of prednisone (or equivalent) for ≥ 3 months’ duration if they are in the following categories [4,5]:
There are currently no evidence-based guidelines for the prevention and treatment of steroid-induced osteoporosis in children. Currently experts recommend [4]:
The use of systemic steroids is associated with hypertension, hyperglycaemia and obesity, which are risk factors for ischaemic heart disease and heart failure.
Assess the patient’s current risk factors for cardiovascular disease (CVD); this will include assessing:
The risk of the patient developing CVD can be reduced through the following lifestyle factors:
Calculate the patient’s cardiovascular risk. If the outcome of your calculations presents their risk as:
Assess the patient’s lipid profile at baseline, after 1 month, and then every 6–12 months after the initiation of systemic steroids. The Framingham Risk Score can be used to assess an individual’s 10-year risk of developing CVD, and can be used to guide treatment [4].
The use of systemic steroids has effects on glucose levels within hours, and is associated with hyperglycaemia and insulin resistance [4].
Assess the patient’s blood glucose levels. The recommended ranges are:
Glycosylated haemoglobin (HbA1c) is a measure of blood glucose over the preceding 3 months. The level of HbA1c indicates the risk of diabetes:
If the blood glucose levels are not within the normal ranges, initial management should be the implementation of lifestyle modification strategies as described for CVD. If blood glucose targets are not met, glucose-lowering medications are recommended.
Random plasma glucose > 11 and < 15 mmol/L should be treated with one of the following medications (as with pre-existing type 2 diabetes):
If blood glucose levels are > 15 mmol/L, insulin may be required to control glycaemia.
Steroid-induced diabetes in children will require advice from a multidisciplinary paediatric diabetes healthcare team. Lifestyle interventions are first-line therapy, followed by insulin [4].
Systemic corticosteroids can cause an increased risk of gastrointestinal events, such as gastritis, peptic ulcer, and gastrointestinal haemorrhage [1,4].
Assess the patient’s history of risk factors for peptic ulcer [5]; these include:
Following a thorough assessment, if you think a patient is at high risk of gastrointestinal disease, consider prescribing a H2 receptor antagonist or a proton-pump inhibitor [4].
The use of systemic steroids directly affects immune cells, such as T-lymphocytes, and the immune system cannot respond to infection as effectively as normal, leading to an increased risk of infection [4].
Encourage the patient to present to you early if they are unwell.
Take an immunisation history, covering the patient’s routine childhood and adult vaccines, before initiating therapy; routine vaccines include [9]:
Any testing for chronic infection should include:
Infection can be minimised by frequent hand washing, avoiding exposure to others with infection, and the use of appropriate vaccines.
Give missing or indicated vaccines before initiating systemic steroid therapy. Give live vaccines at least 2–4 weeks beforehand, as systemic steroids reduce the efficacy and increase the risk associated with vaccines [5].
Avascular necrosis occurs when there is loss of blood flow to the bone, leading to infarction of the bone, often at the femoral head. The risk of developing osteonecrosis increases with increased doses of systemic corticosteroids [5].
Assess the patient’s previous medical history for risk factors for avascular necrosis; risk factors include:
Ask about the patient’s alcohol consumption, as this can increase risk of avascular necrosis. Recommend they avoid excessive alcohol consumption.
Monitor patients with an elevated risk of avascular necrosis closely.
Monitor any joint pain and the range of motion in the joint as early diagnosis and appropriate intervention can prevent or delay the progression of avascular necrosis [1].
If avascular necrosis is suspected, you can diagnose using:
Treatment of avascular necrosis initially involves:
If conservative management fails, additional options can be considered, such as surgery [1].
The use of systemic steroids may alter the patient’s mood or cause memory deficit, agitation, anxiety, lethargy and psychosis [1,4].
The patient must be screened for any of the following risk factors:
Low serum albumin levels are a predictor of psychosis in patients with systemic lupus erythematosus [1,4].
Monitor any changes in the patient’s behaviour or cognitive status.
Lithium carbonate can be used as prophylaxis for psychosis in those patients who are predisposed to psychosis [1,4].
Persistent symptoms of psychosis may indicate antipsychotic therapy is required. Most patients recover with either dose reductions or cessation of systemic steroids.
If a patient experiences sleep disturbances and unpleasant dreams, consider changing their dose of systemic steroids to a single morning dose, or administer drugs with sedative effects at night [4].