Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Introduction Uses Contraindications Medications Benefits Side effects and risks
An intralesional injection is the direct injection of a therapeutic substance into a lesion or into the skin. The aim of an intralesional injection is to deliver a high concentration of medicine into the site of the pathology to maximise efficacy while minimising systemic adverse effects of the drug.
The most common medication used for intralesional injection is a corticosteroid such as triamcinolone acetonide.
Intralesional steroid is used for localised inflammatory, hyperplastic, and hypertrophic lesions [1,2].
Other drugs that are sometimes injected into skin lesions include 5-fluorouracil (eg, for keloid scar) [3], bleomycin (eg, for viral wart) [4], methotrexate (eg, for nail psoriasis) [5], rose Bengal, and interferon (see Topical and intralesional immunotherapy for melanoma metastases).
Cosmetic use of injected substances include:
The intralesional injection should not be used in or around an active bacterial skin infection (impetigo, atypical mycobacteria), fungal infection, or viral infection (herpes simplex).
It should not be used if the patient is allergic to the specific drug or excipient [6].
Intralesional injections may not be suitable for widespread or extensive lesions.
Triamcinolone acetonide is widely available with 10 mg/mL or 40 mg/mL strengths marketed in Australasia. The dose depends on the type, size, and location of the lesion.
0.1–0.2 mL is injected per square centimetre of involved skin. The steroid can be diluted with normal saline or plain local anaesthesic.
The medicine is placed in the dermis if it is a flat lesion (eg, alopecia areata), or within the lesion, if it is raised (eg, hypertrophic scar).
A Luer-lock syringe (figure 1) or an insulin syringe can be used with a 25 to 30-gauge needle, or a special syringe set with a controlled depth needle can be used (eg, Dermojet). Otherwise, the medicine may spill if the needle disconnects from the syringe due to the resistance of dense collagen tissue. Insert the needle at a 10 to 20-degree angle for a flat lesion (figure 2), or at an increased angle for a raised lesion. Subcutaneous injection should be avoided.
The procedure can be repeated every 4–6 weeks for 2–3 cycles or longer.
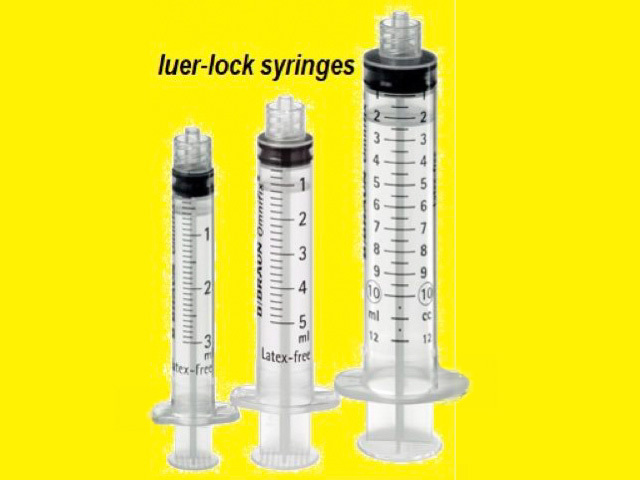
Figure 1: Luer-lock syringes
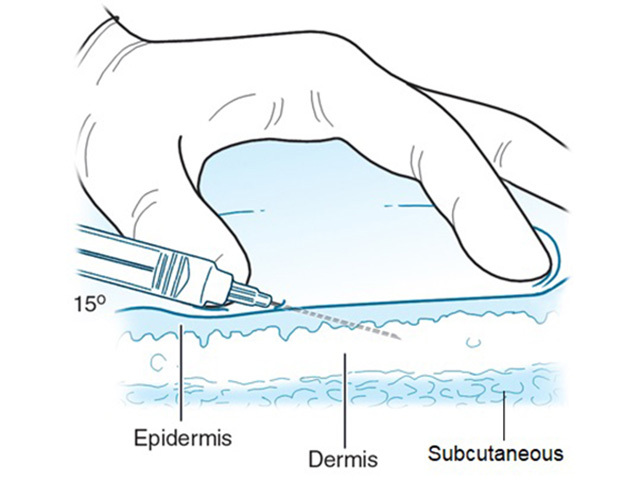
Figure 2: injecting into the superficial dermis
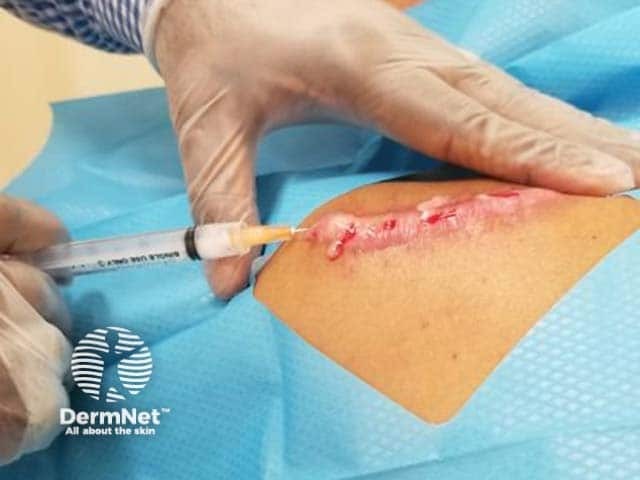
Figure 2 Credit: McGraw Hill Education; used with permission.
An intralesional injection is a painful procedure even when mixed with local anaesthetic, as numbing occurs a few minutes later. Tips for minimising pain are:
An intralesional injection is a simple office-based and relatively safe procedure with proven benefits in specific skin conditions.
The possible side effects of intralesional injection of a corticosteroid are:
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).