Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Aamenah Al-Ani, Medical Student, University of Auckland, Auckland, New Zealand. DermNet Editor in Chief: Adjunct A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand. January 2018.
Introduction
Demographics
Causes
Clinical features
Diagnosis
Differential diagnoses
Treatment
Follow-up
Outcome
Amelanotic melanoma is a form of melanoma in which the malignant cells have little to no pigment. The term 'amelanotic' is often used to indicate lesions that are only partially devoid of pigment while truly amelanotic melanoma where lesions lack all pigment is rare.
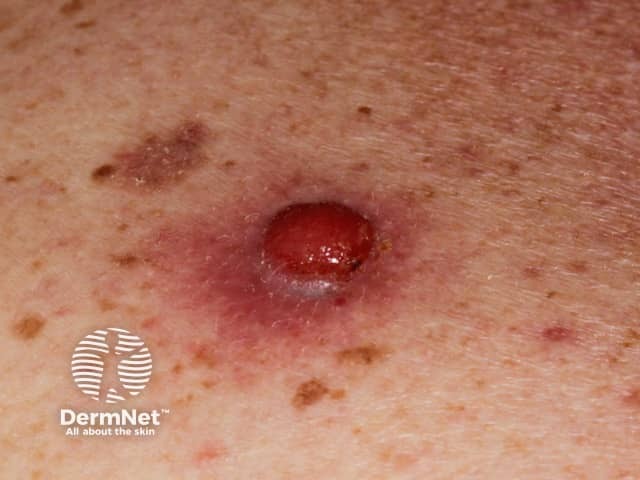
Nodular melanoma
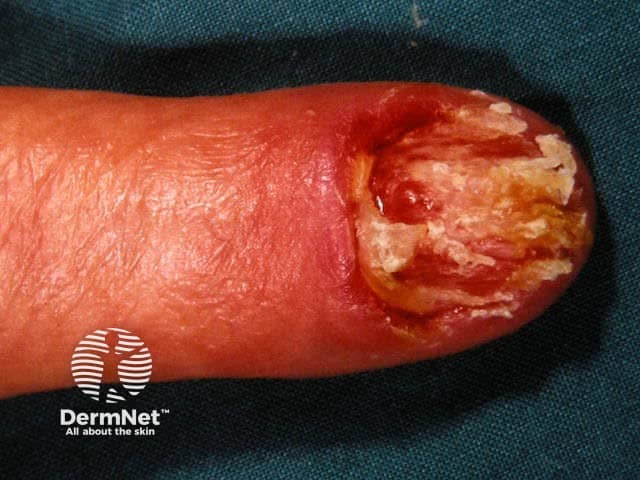
Ungual melanoma
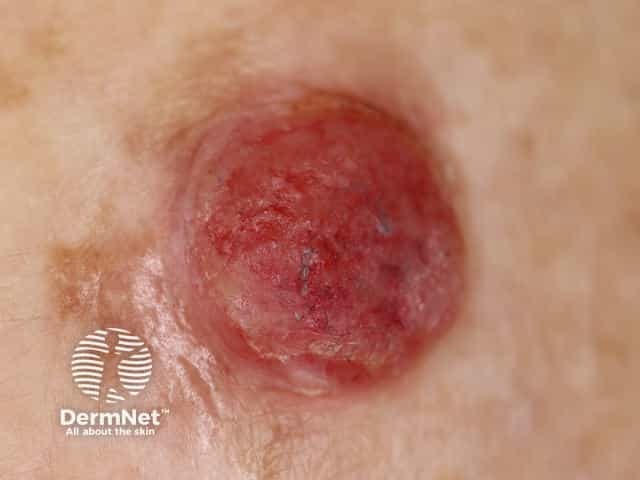
Amelanotic melanoma 3mm
See more images of amelanotic melanoma
Amelanotic melanoma accounts for approximately 2–20% of all melanomas. The incidence of truly amelanotic melanoma is difficult to estimate, given that many hypopigmented lesions are labelled as amelanotic.
Risk factors for developing amelanotic melanoma include:
Melanoma is caused by malignant melanocytes. The development of malignancy in melanocytes is due to genetic changes in DNA, but how and why this occurs is largely unknown. The melanoma cells in amelanotic melanoma cannot produce mature melanin granules, which results in lesions that lack pigment.
The lack of pigment may be due to:
Amelanotic melanomas are classically described as 'skin coloured'. A significant proportion is red, pink, or erythematous. Typical early lesions present as asymmetrical macular lesions that may be uniformly pink or red and may have a faint light tan, brown, or grey pigmentation at the periphery. The borders may be well- or ill-defined.
Any subtype of melanoma can be amelanotic.
Amelanotic melanomas may not display the clinical ABCDE criteria (Asymmetry, Border irregularity, Colour variation, large Diameter) that are classically used as melanoma warning signs. Patients and clinicians may not be alert to suspect non-pigmented lesions as melanoma and so amelanotic melanomas are often misdiagnosed. Expanding the ABCD warning signs to include the 3 R’s (Red, Raised, Recent change) may help screen for amelanotic melanoma.
Amelanotic melanomas have little to no pigment and can be clinically challenging to diagnose.
A patient’s history of observing a change in a lesion is an important diagnostic factor with amelanotic lesions and amelanotic melanoma should be considered in the differential diagnosis.
Examination of the entire skin surface is also important, as sun damage (eg, actinic keratoses) and other pigmented lesions may provide clinical clues that a non-pigmented or hypopigmented lesion may be an amelanotic melanoma.
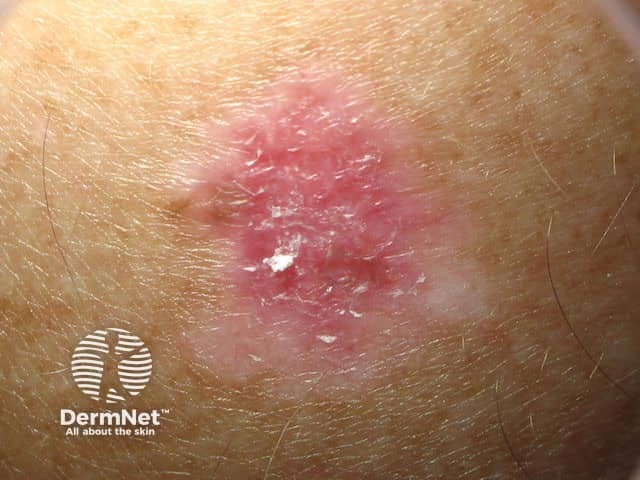
Amelanotic superficial spreading melanoma in situ
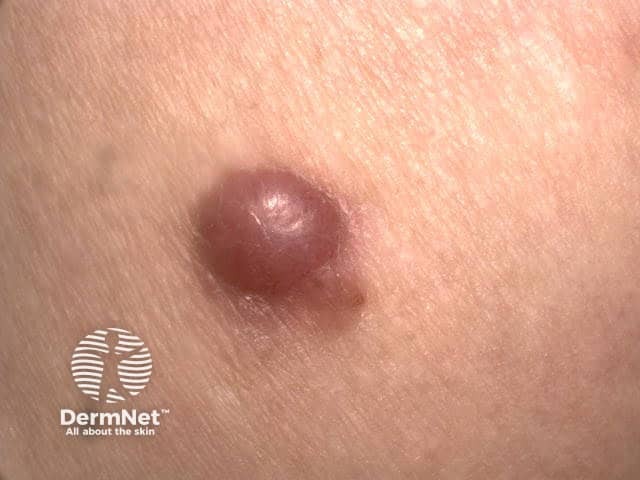
Amelanotic nodular melanoma
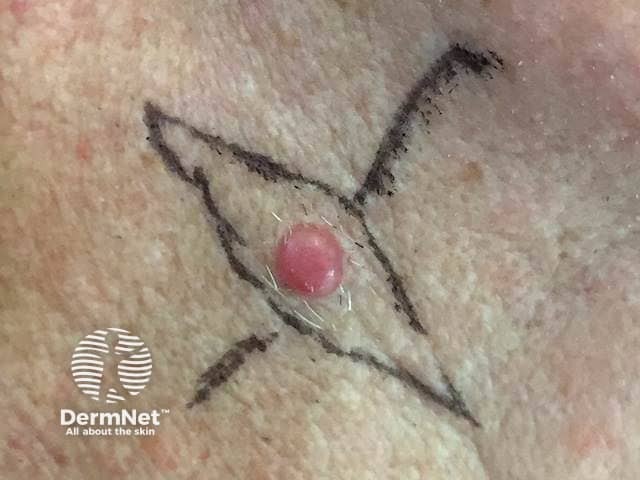
Amelanotic nodular melanoma
Dermoscopy, the examination of a lesion using a dermatoscope, can be helpful in the diagnosis of suspicious lesions. Vessel morphology should be evaluated carefully if lesions are lacking pigmentation.
The characteristic dermoscopic features of amelanotic melanoma are:
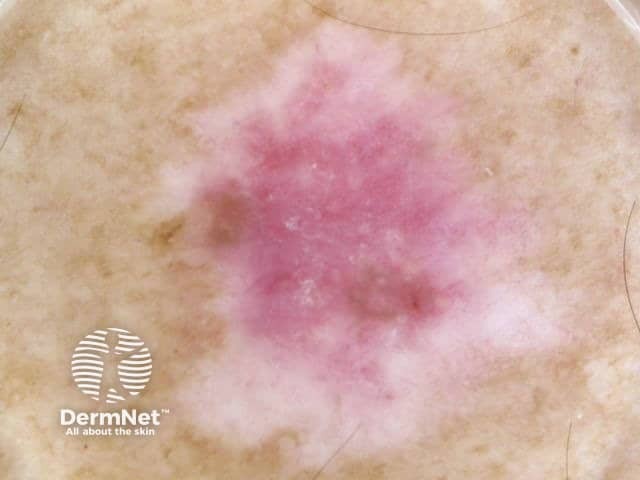
Superficial spreading melanoma in situ
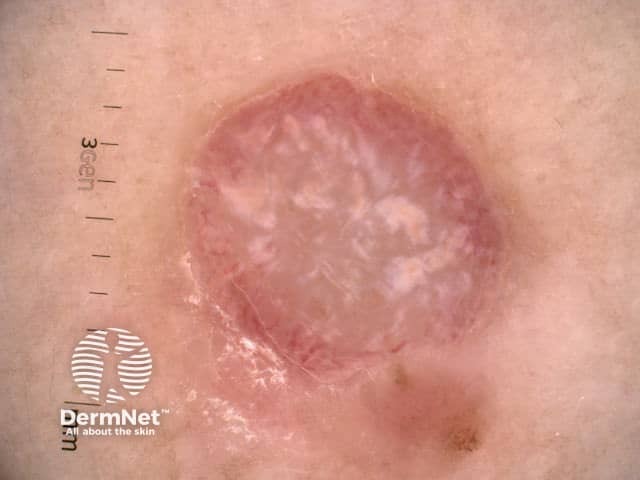
Nodular melanoma
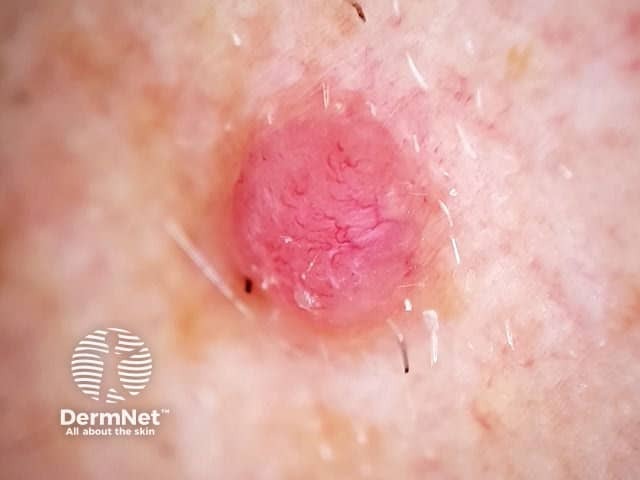
Nodular melanoma
Lesions that are suspicious of amelanotic melanoma should be excised with a 2–3 mm clinical margin and sent for pathological diagnosis (excision biopsy).
Amelanotic melanoma can have varied histopathological appearances. Amelanotic melanoma can masquerade as a number of non-melanocytic neoplasms.
The pathologist’s report features both a macroscopic and microscopic description of the excised lesion and includes:
The report may also include comments about the cell type and its growth pattern, invasion of blood vessels or nerves, inflammatory response, regression, immunohistochemistry, and whether there is any associated original benign melanocytic lesion.
The Breslow thickness is reported for invasive melanomas. It is the vertical measurement in millimetres from the top of the granular layer (or base of superficial ulceration) to the deepest point of tumour involvement. The Breslow thickness is the single most important local prognostic factor in primary melanoma, as thicker melanomas are more likely to metastasise (spread).
The Clark level indicates the anatomic plane of invasion. This is useful for predicting outcome in thin tumours and less useful for thicker ones in comparison to the value of the Breslow thickness.
Clinically, amelanotic melanoma may present as an erythematous scaly macule, plaque, or nodule with irregular borders, mimicking numerous other skin lesions such as:
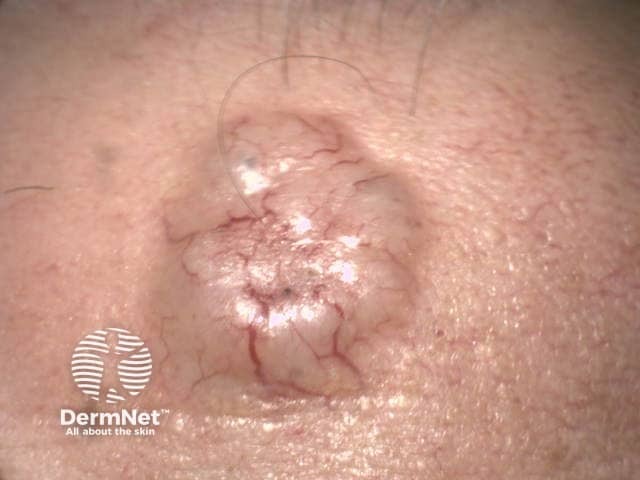
Nodulocystic basal cell carcinoma
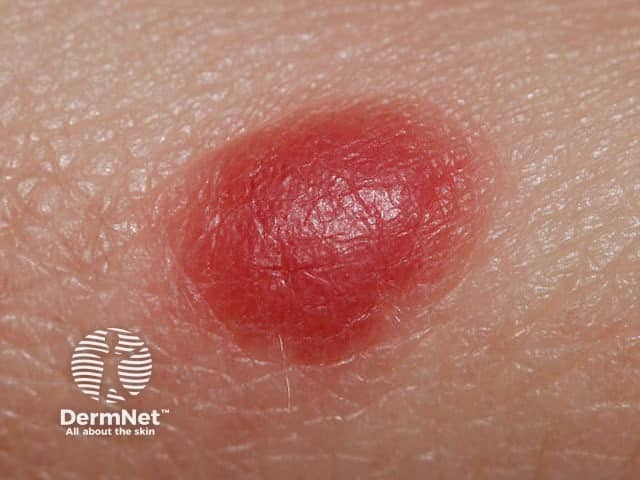
Spitz naevus
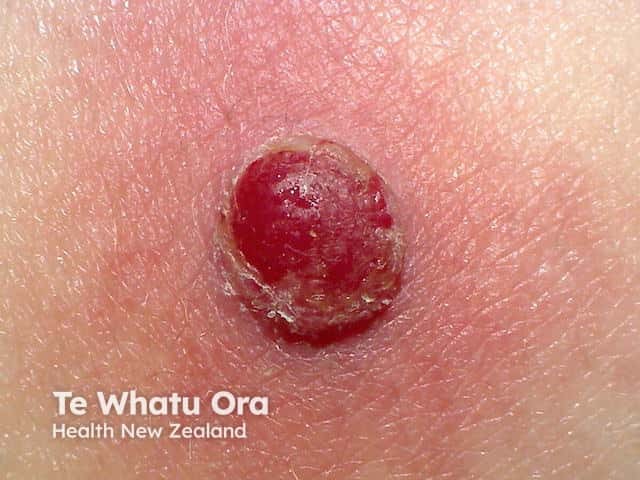
Pyogenic granuloma
Amelanotic melanoma is treated in the same way that a pigmented melanoma is treated.
After diagnostic excision, the next step is wide local excision of the wound with a 10–20 mm margin of normal tissue. The extent of surgery depends on the Breslow thickness of the melanoma and its site. Amelanotic melanomas may be incompletely excised despite the recommended margins as the margins are often difficult to define. A comprehensive histological examination including immunohistochemical staining helps determine the edge of a tumour. Further re-excision may be necessary. The recommended margins in the provisional 2013 Standards of service provision for melanoma patients in New Zealand are as follows:
Sentinel lymph node biopsy may be discussed with patients with melanomas thicker than 0.8 mm. This can provide staging and prognostic information.
Amelanotic melanoma can metastasise (spread to distant sites such as lymph nodes or elsewhere in the body). These cases require individualised treatment that may include surgery, radiotherapy, chemotherapy, or targeted therapy.
Follow-up is an important part of the management for amelanotic melanoma because it allows an opportunity to detect recurrences early. It also allows an opportunity to detect new primary melanomas at the earliest opportunity. A second invasive melanoma occurs in 5–10% of patients and an unrelated melanoma in situ affects more than 20% of patients.
The 2008 Clinical practice guidelines for the management of melanoma in Australia and New Zealand recommend the following for the follow-up of patients with invasive melanoma:
The prognosis of amelanotic melanoma is similar to that of pigmented melanomas. Prognostic factors include the Breslow thickness of the melanoma at the time of excision (this is considered to be the most important factor), the location of the lesion, patient age, and sex. Importantly, because of their atypical clinical features, amelanotic melanomas may have a delay in their diagnosis and, consequently, are often more advanced than pigmented melanomas when diagnosed.
The risk of metastasis is directly related to the Breslow thickness, with thicker melanomas being more likely to metastasise. The 2008 Clinical practice guidelines for the management of melanoma in Australia and New Zealand report that metastases are rare for thin melanomas (< 0.75 mm), with the risk increasing to 5% for melanomas 0.75–1.00 mm thick. Melanomas thicker than 4.0 mm have a significantly higher risk of metastasis of 40%.