Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: George Zhang, Medical Student, University of Auckland. DermNet New Zealand Editor in Chief: Hon A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand. Copy editor: Maria McGivern. April 2017.
Introduction Diagnosis Biopsy site Transport and storage of the biopsy sample Laboratory procedures Interpretation of results Reliability
Direct immunofluorescence (DIF) is a technique used in the laboratory to diagnose diseases of the skin, kidney, and other organ systems. It is also called the direct immune fluorescent test or primary immunofluorescence.
DIF involves the application of antibody–fluorophore conjugate molecules to samples of patient tissue obtained from biopsies. These antibody–fluorophore conjugates target abnormal depositions of proteins in the patient’s tissue. When exposed to light, the fluorophore emits its own frequency of light, seen with a microscope. The particular staining pattern and type of abnormal protein deposition seen in the tissue sample help diagnose the disease.
DIF is useful in the diagnosis of suspected autoimmune disease, connective tissue diseases and vasculitis. The staining patterns seen in tissue samples may be specific to a disease entity or they may need to be interpreted with the clinical and histological findings.
DIF is useful in diagnosing the following autoimmune bullous diseases:
DIF is useful in diagnosing the following connective tissue diseases:
DIF is useful in diagnosing cutaneous vasculitis and some other types of inflammatory skin disease:
The optimal site for taking a biopsy of the skin depends on the suspected disease.
Another biopsy is usually taken for routine haematoxylin–eosin (H&E) histology. Note that the optimal biopsy site and timeframe required differs for these examples.
Specimens for DIF should not be placed in formalin, as this alters proteins and significantly diminishes the accuracy of results. Transport the specimen to the laboratory as soon as possible. Options for transporting the sample include:
Each laboratory has its own protocols. Specimens in saline may yield superior sensitivity over other media if processed within 24–48 hours.
DIF may be carried out in an automated machine or manually. The process involves first making frozen sections then carrying out immunofluorescence.
Punch biopsy on saline-soaked gauze

Specimen in gel

Liquid nitrogen storage

Frozen specimen

Specimen in liquid nitrogen

Cutting thin slices of skin

Five slides are made

A special pen is used

Slides washed in solution

Making up reagents

The reagents

Reagent dropped on slides

Wash in solution

Prepare for cover slip

Glass cover
The prepared immunofluorescence slides are examined by a pathologist to determine the primary sites of immune deposition (if any), the classes of immunoglobulin or other immune deposits, and the patterns of deposition. Staining patterns can be classed into five groups:
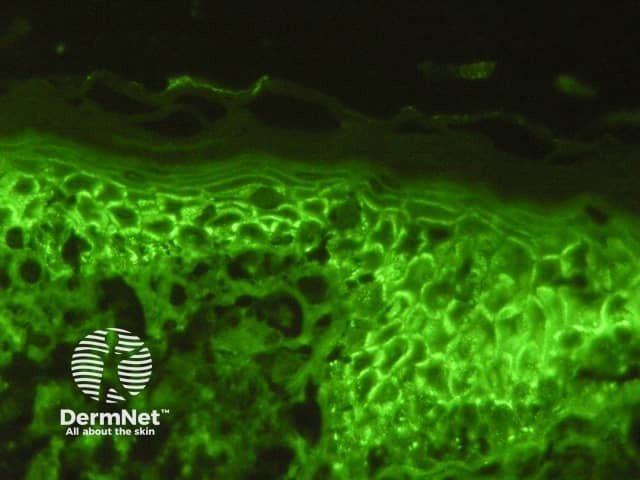
Pemphigus vulgaris
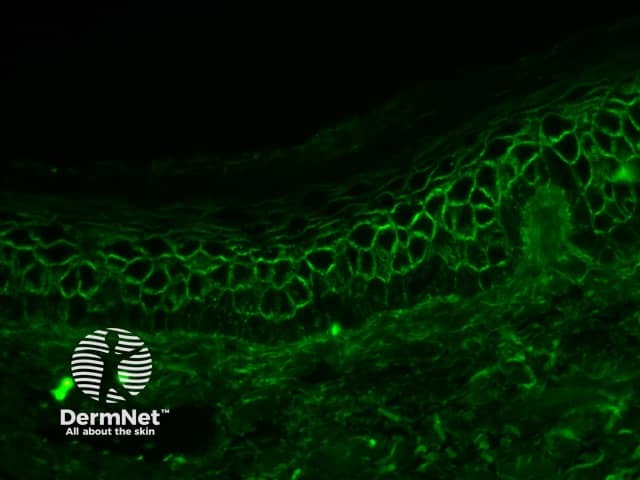
Pemphigus foliaceus
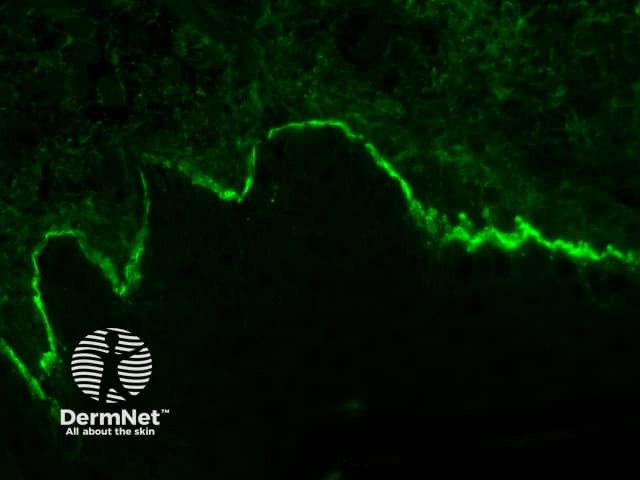
Bullous pemphigoid
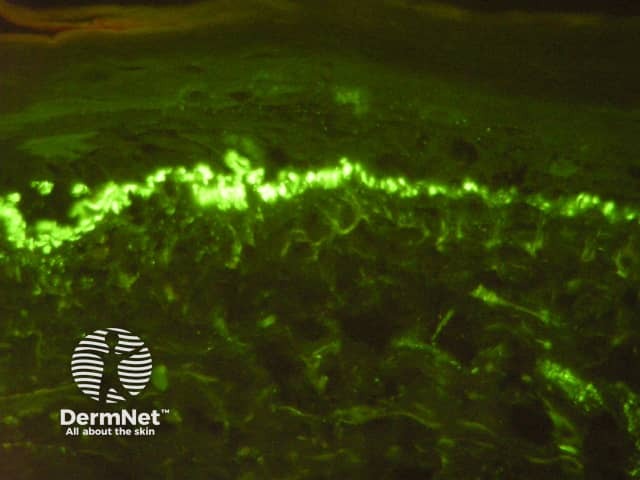
C3 on basement membrane zone
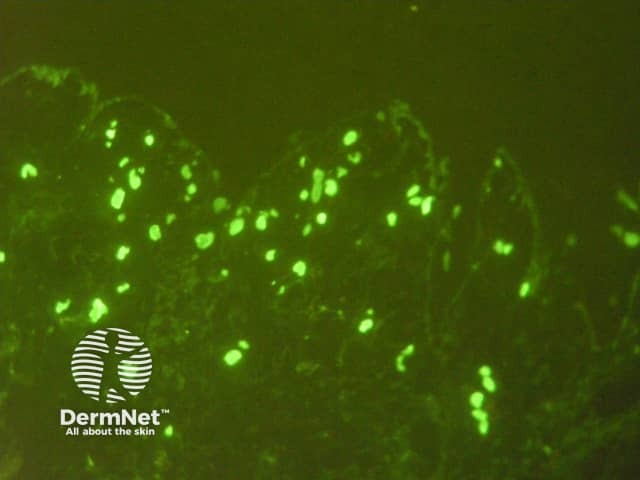
Vascular staining
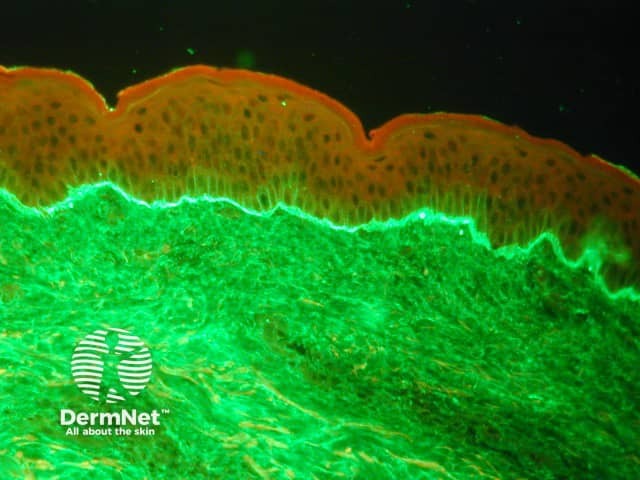
Linear IgA bullous dermatosis
DIF testing is moderately sensitive in detecting a range of diseases. The sensitivity for DIF approaches 100% for the pemphigus group of diseases, and sensitivity has been reported to be 55–96% for bullous pemphigoid. A study of ten patients with Henoch–Schönlein purpura and nine with lupus erythematosus demonstrated positive DIF testing in all of the patients.
The number of false-negative results with DIF depends on the quality of the sample, laboratory processing, and whether or not the patient has been on treatment. Reasons for false-negative results include:
Cutaneous immunofluorescence testing — Mayo Medical Laboratories