Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Autoimmune/autoinflammatory
Author(s): Dr Alpana Mohta, Dermatologist; and Dr Achala Mohta, PCMO, Sardar Patel Medical College, India (2023)
Previous contributors: Dr Anoma Ranaweera, PhD in Clinical Biochemistry; and Dr Amanda Oakley, Dermatologist (2014)
Reviewing dermatologist: Dr Ian Coulson
Edited by the DermNet content department
Introduction
Mechanism
Uses
Precautions and contraindications
Pre-treatment considerations and monitoring
Side effects and risks
Rituximab is a biologic medicine used primarily to treat B-cell lymphoma. Recently, it is has also shown to be useful in the treatment of several severe skin diseases. In 2018, the FDA approved its use for moderate to severe pemphigus vulgaris, an immunobullous disease of the skin.
Rituximab is a monoclonal antibody directed against the CD20 antigen on the surface of normal and malignant B lymphocytes. Rituximab has human and mouse-derived components. Given by intravenous injection, it is very effective at destroying normal and malignant B lymphocytes that are carrying the CD20 antigen. However, it can have severe side effects.
Trade names include Rituxan® and MabThera®.
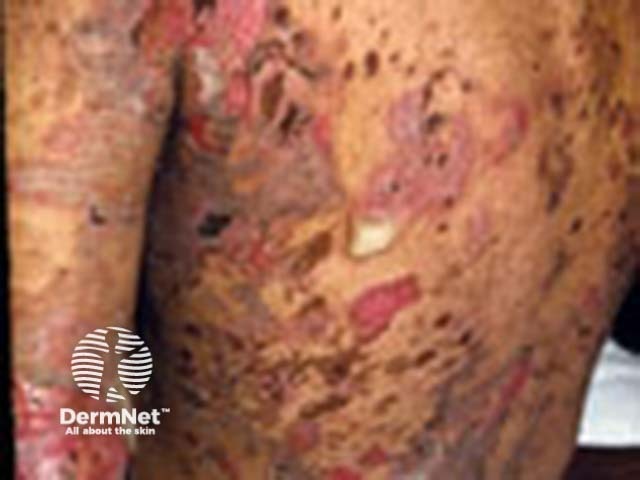
Severe pemphigus vulgaris; rituximab is indicated
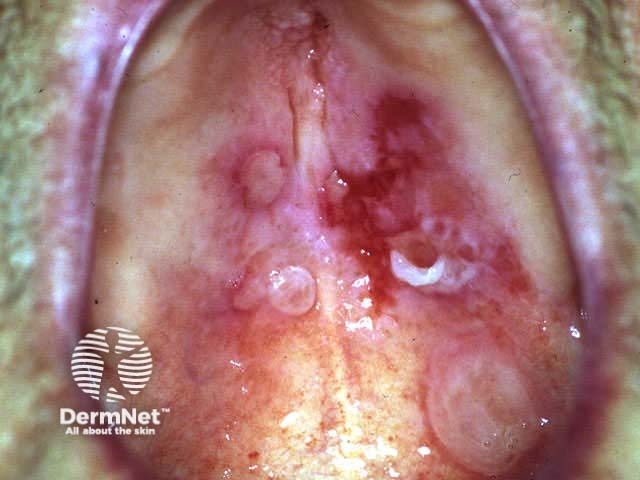
Severe oral cicatricial pemphigoid unresponsive to steroids and dapsone; rituximab is indicated.
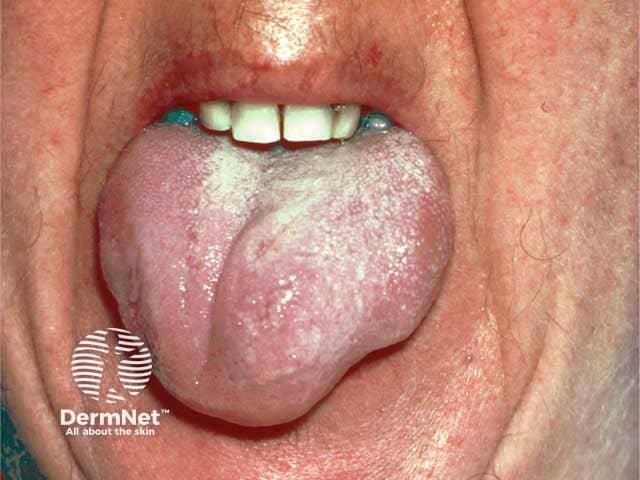
Angioedema due to acquired C1esterase deficiency and an underlying lymphoma; it responded to rituximab
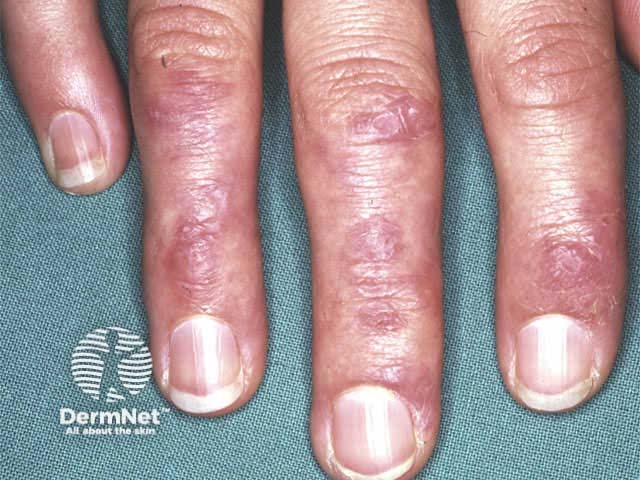
Severe dermatomyositis of the hands unresponsive to steroids and methotrexate; rituximab is one of several therapeutic options
B lymphocytes are a type of white blood cell. Their role in immune reactions includes:
Rituximab is an immunoglobulin G1 (IgG1) kappa monoclonal antibody composed of a murine (mouse) variable region (Fab portion) that is fused onto a human constant region (Fc portion). The Fab portion binds to the CD20 antigen on the surface of pre-B and mature B lymphocytes. The Fc portion then recruits immune cells that destroy these lymphocytes. Mechanisms may include:
The exact contribution of each mechanism remains unclear, and different mechanisms may prevail in various diseases.
Rituximab has a limited effect on other immune cells.
Rituximab is indicated for the following conditions:
Rituximab has also been found to be useful in a variety of other immune-mediated and autoimmune disorders in which traditional therapy has failed or resulted in side effects.
It has been reported to be helpful (off-label) for:
To date, rituximab has been effective in some cases of the following skin conditions:
The most suitable dose of rituximab for these disorders has not yet been determined.
Cutaneous B-cell lymphoma includes follicle centre, marginal zone, and diffuse large B-cell lymphoma, which have all been reported to respond to intravenous rituximab. Direct injections into the skin lesions have also been successful, allowing a lower dose to be used. However, after the initial response, the lymphoma may recur within several months. Retreatment may or may not prove successful as lymphomas can lose CD20 expression and, with that, susceptibility to rituximab.
Primary blistering diseases (also called autoimmune bullous disorders) are associated with autoantibodies directed against various structural support proteins in the epidermis and dermoepidermal junction.
Rituximab has been successfully used for pemphigus vulgaris and treatment-resistant cases of:
Dermatomyositis is an autoimmune disease characterised by inflamed, weakened muscles associated with a characteristic rash. How this occurs is unknown, but it involves T cells, B cells, and antibodies directed against the endothelial cells lining blood vessels in the muscles.
Treatment of treatment-resistant dermatomyositis with rituximab has led to the improvement of muscle and skin disease in several patients. Clinical trials have been set up to determine the role of rituximab in dermatomyositis.
Chronic graft versus host disease (GVHD) affects 60–70% of long-term survivors after bone marrow transplantation. It results in lichenoid and sclerodermatous skin changes as well as disease of internal organs. GVHD often fails to respond to conventional treatment, and a quarter of affected patients die from it.
Chronic GVHD has been thought to be due to donor T cells, but drugs targeting T cells have not proved very useful. There is now mounting evidence that B cells are responsible. There have been reports of improvement with rituximab in some patients with GVHD.
Vasculitis is classified as a type-III hypersensitivity reaction involving immune complexes, that is, antibodies bound to antigens in the affected blood vessels. Exactly how rituximab works in vasculitis is unknown.
In April 2011, the US FDA approved rituximab for antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (granulomatosis with polyangiitis and microscopic polyangiitis). Case reports suggest rituximab is also efficacious in cutaneous vasculitis due to cryoglobulinaemia.
Atopic dermatitis (eczema) results from a complex interaction between various immune cells and proteins and in many patients is characterised by high levels of serum IgE. B cells and plasma cells produce IgE, so theoretically, B-cell depletion could help treat the disease.
Rituximab has resulted in an impressive improvement of atopic dermatitis in some patients with severe disease. However, although researchers confirmed a reduction in circulating B cells in their patients, they found little change in IgE levels.
Rituximab has been successfully used to treat refractory cutaneous lupus erythematosus.
Current safety information is based on treatment for non-Hodgkin lymphoma and rheumatoid arthritis and may not be applicable when rituximab is used for other disorders.
Though there have been no formal drug interaction studies with rituximab, concomitant use with cisplatin should be avoided as this combination has been associated with renal failure.
Prior to the rituximab infusion, premedication is administered 30 minutes in advance. This usually includes paracetamol and an antihistamine and may include glucocorticoids.
Determining the optimal dosage and regimen of rituximab for pemphigus treatment remains a topic of ongoing discussion. Various therapy approaches are currently employed, including the following protocols.
Rituximab is given by IV infusion. The total infusion duration is approximately 5 to 6 hours, depending on the infusion rate.
Most patients experience mild-to-moderate side effects from their first infusion of rituximab. The most common symptoms are:
The symptoms usually settle if the infusion is stopped temporarily and tend to be less severe with subsequent infusions. Pre-treatment with paracetamol and antihistamines is recommended. A corticosteroid may also be used.
Although infections are rare, some patients have died after rituximab infusions due to:
Patients should be urged to seek prompt medical attention if they develop new neurological symptoms such as changes in vision, balance, or thought processes.
Other deaths from rituximab have been due to:
Many other less severe adverse reactions have been reported, including a variety of rashes and:
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).