Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author(s): Dr Lachlan Dat Wah Lau, Dermatology Resident; Dr Mi Vu, Dermatologist; and A/Prof Laura Scardamaglia, Dermatologist, Melbourne, Australia (2023).
Previous contributors: Vanessa Ngan, Staff Writer (2006).
Reviewing dermatologist: Dr Ian Coulson
Edited by the DermNet content department
Introduction
Demographics
Causes
Clinical features
Variation in skin types
Complications
Diagnosis
Differential diagnoses
Treatment
Prevention
Outcome
Drug-induced photosensitivity (DIP) is a common adverse drug reaction resulting in a cutaneous eruption after exposure to visible or ultraviolet (UV) radiation in patients taking topical or systemic photosensitising medications.
There are many established and new drugs with photosensitising potential. The drug or its metabolites present within the skin absorbs UV radiation and triggers a chemical photosensitivity reaction, either phototoxic (more common) or photoallergic.
Photosensitising properties are sometimes used for therapeutic purposes in photodynamic therapy and photochemotherapy.
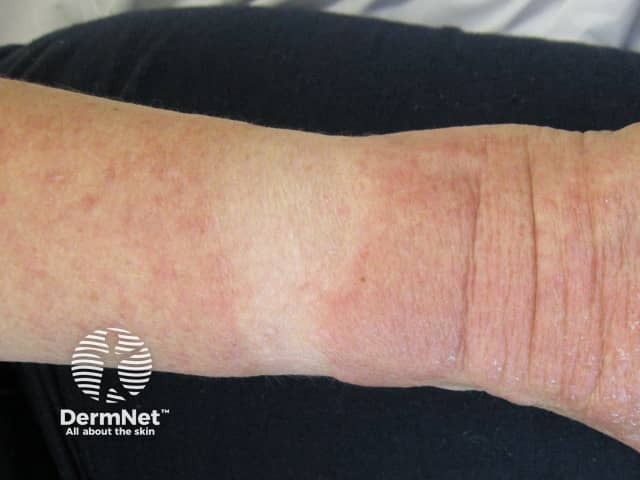
A photosensitive drug eruption showing sparing under the watch and strap
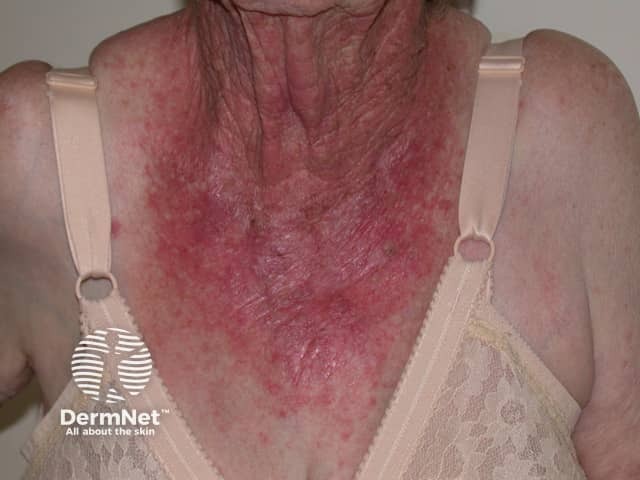
A phototoxic drug eruption due to an oral non-steroidal anti-inflammatory drug
Click here for more images of drug-induced photosensitivity
Drug-induced photosensitivity (DIP) is not genetically inherited, and there is no known age, gender, or racial predilection to developing a photosensitising cutaneous eruption.
DIP is generally considered to account for up to 8% of reported adverse cutaneous reactions to drugs, although it is likely underdiagnosed and underreported.
Phototoxic reactions are more common than photoallergic reactions.
Phototoxic drugs |
Photoallergic drugs |
|||
Antibiotics |
Fluoroquinolones (eg, ciprofloxacin) |
Fluoroquinolones |
||
Antifungals |
||||
Cardiovascular and diuretic agents |
Amiodarone |
Quinidine |
||
Furocoumarins / psoralens eg, for PUVA (photochemotherapy) |
Methoxsalen (8-methoxypsoralen) |
Topical psoralens |
||
Ibuprofen |
Diclofenac |
Celecoxib |
Diclofenac |
|
Other agents |
BRAF inhibitors (eg, vemurafenib) |
Antineoplastics (eg, fluorouracil cream |
Pyridoxine |
Fragrances (eg, musk ambrette, 6-methylcoumarin) |
A phototoxic reaction is a non-immunologic cutaneous reaction that appears acutely within minutes to hours on sun-exposed skin after taking photosensitising medications.
Phototoxicity could theoretically occur in any patient taking photosensitising medication. However, there is a dose-dependent relationship where it frequently requires a high dose of medication or light exposure that exceeds a critical threshold.
Phototoxic skin damage begins when the drug or its metabolite within the skin absorbs ultraviolet radiation (UVR) or visible light. Then, photochemical pathways may involve:
The resulting oxidative stress due to the formation of ROS, and direct cellular damage caused by the free radicals, manifests as an exaggerated sunburn-like reaction.
A photoallergic reaction is an immune-mediated delayed type IV hypersensitivity reaction. The key sensitising event involves the photobinding of a drug (or its metabolite) to skin biomolecules upon exposure to UVR; this forms a photoallergen. The initial sensitisation period typically takes 7–10 days. Upon subsequent re-exposure to light, an eczematous or lichen planus-like eruption that may spread beyond sun-exposed areas occurs 24–72 hours later.
The clinical features of DIP depend on the specific photosensitising agent involved and the type of skin reaction it triggers (phototoxic or photoallergic).
Phototoxicity |
Photoallergy |
|
|
|
|
Characterised by erythema, oedema, a burning and stinging sensation, with vesicles and bullae in severe reactions |
|
|
|
|
|
|
|
Click here for drug-induced photosensitivity images
Phototoxicity is more commonly observed in individuals with lighter skin types. It is believed that increased melanin pigment provides protection due to its antioxidant properties against free radicals. The only known exception is a diltiazem-associated photodistributed hyperpigmentation reaction that is more commonly seen in patients with darker skin types.
Photoallergic reactions occur equally across different skin types.
DIP is a clinical diagnosis that can be supported with reproducibility on phototesting.
History |
|
Examination |
|
Investigations |
|
Discontinuing the photosensitising medication and sun protection remain the mainstay preventative measures for DIP.
DIP often resolves upon ceasing the causative photosensitising medication, although postinflammatory hyperpigmentation may persist in phototoxic reactions. In cases when discontinuing the medication is not an option, symptom control with strict sun protection can effectively manage DIP symptoms.
Uncommonly, in photoallergic contact dermatitis, chronic exposure to photosensitising drugs can lead to photosensitivity that persists for months or years after the medication has been ceased.
The role of photosensitising medications in photocarcinogenesis is debated and not yet fully elucidated.