Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Dr Anes Yang, Clinical Researcher, Department of Dermatology, St George Hospital, Australia. Dr Monisha Gupta, Consultant Dermatologist, Department of Dermatology, Liverpool Hospital, Australia. DermNet Editor in Chief: Adjunct A/Prof Amanda Oakley, Dermatologist, NZ. January 2018.
Introduction
How it works
Anti-inflammatory effects
Antiproliferative and immunomodulatory effects
Pharmacokinetics
Common uses
Other uses
Contraindications
Monitoring
Side-effects and risks
Drug interactions
Pregnancy
Antimalarial medications are drugs with immunomodulatory and anti-inflammatory effects. They are used to treat various skin conditions.
Antimalarial medications used in dermatology include:
Hydroxychloroquine has largely replaced quinacrine and chloroquine due to its better safety profile [1].
Antimalarials are:
Antimalarial drugs reduce the production of cytokines that induce an inflammatory response by activating macrophages, dendritic cells, and lymphocytes where they are trapped within cytoplasmic lysosomes (enzyme-filled organelles that dissolve molecules) [2].
The anti-proliferative and immunomodulatory effects are mediated by:
Antimalarial medications prevent platelet aggregation and act as prostaglandin antagonists due to inhibition of phospholipase A2 [3]. It is unclear whether the antithrombotic effect is therapeutically useful. Hydroxychloroquine has been associated with a 15–20% decrease in serum cholesterol, triglyceride, and low-density lipoprotein levels [1]. It reduces glucose by decreasing insulin degradation resulting in lower levels of glycated haemoglobin (HbA1c). It is antiviral, antineoplastic, and may improve bone density.
Chloroquine and hydroxychloroquine are chemically similar and are part of the amino-quinoline family.
There is no cross-reactivity between the 4-aminoquinolines and quinacrine due to differences in the chemical structure; therefore, an adverse reaction to an aminoquinoline does not preclude the use of quinacrine.
Antimalarial drugs are first-line medications for:
They are used second-line to treat:
Antimalarials are sometimes used to treat other inflammatory skin conditions.
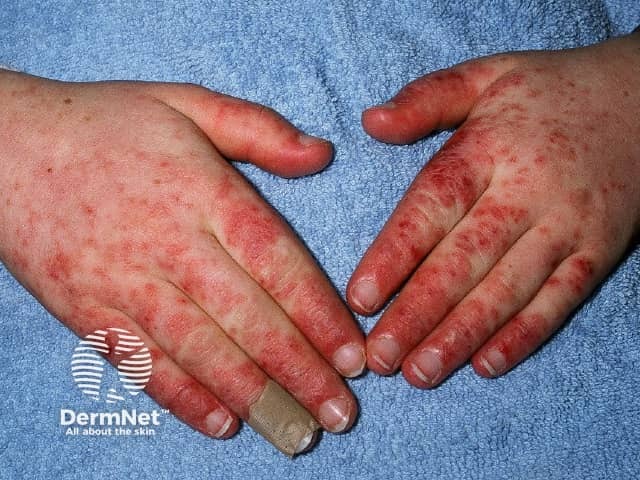
Systemic lupus erythematosus
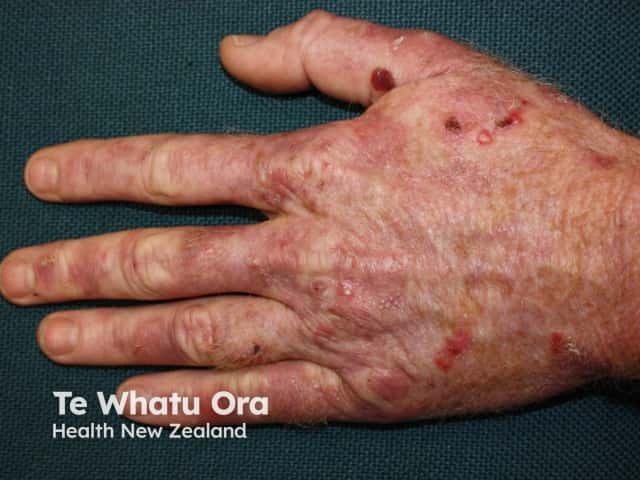
Porphyria cutanea tarda
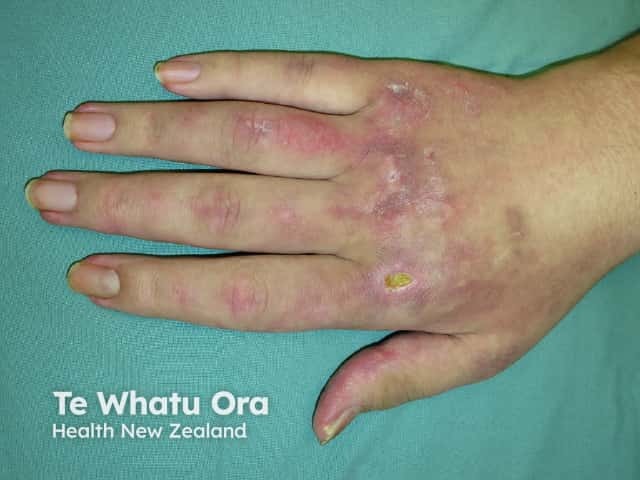
Dermatomyositis
Other inflammatory skin conditions treated with antimalarial drugs include:
Antimalarial drugs should not be used in patients with known hypersensitivity to the drug or pre-existing retinopathy. Care should be taken with children, the elderly, in pregnant and lactating women, and patients with severe renal impairment.
Smoking has been reported to inhibit the P450 enzyme system, decreasing the efficacy of antimalarial therapy, especially in patients on chloroquine [4]. Patients on antimalarial medications are advised not to smoke.
Hydroxychloroquine levels can be measured in the blood.
Except for severe retinopathy, adverse effects from antimalarial drugs usually resolve when they are discontinued.
Chloroquine, and hydroxychloroquine to a lesser extent can cause irreversible retinal toxicity. The cause is thought to be a high affinity for melanin-containing cells in the retinal pigment epithelium [1]. The risk of toxicity is dependent on several factors.
Major risk factors for retinal toxicity include:
Routine screening is essential because early antimalarial retinopathy does not cause symptoms.
Damage patterns vary with ethnicity.
Antimalarial drugs can cause transient or persisting nausea, flatulence, vomiting or diarrhoea that resolves with the reduction or cessation of medication. Symptoms can be minimised by taking the medication with food.
The cutaneous adverse effects of antimalarial drugs include:
Neuropsychological side effects due to antimalarial medications are rare and usually occur in patients treated at higher doses than those used in dermatology. Psychosis, irritability, depression, insomnia, and nightmares have been reported. Antimalarial medications have rarely been known to induce a seizure in predisposed individuals.
Haematological side effects are uncommon with antimalarial drugs. Haemolysis in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, aplastic anaemia, and leukopenia have been reported but are rare [6]. Screening for G6PD deficiency is not routinely recommended for hydroxychloroquine but is required for chloroquine.
Anti-malarial medicines can cause several interactions with other medications [2].
Antimalarial drugs increase the plasma levels of:
There are synergistic antiarrhythmic effects with chloroquine and amiodarone.
Antimalarials decrease the bioavailability of penicillin.
There is increased bioavailability of antimalarial agent with:
There is decreased bioavailability of the antimalarial agent with:
There is an increased risk of myopathy with antimalarials and:
Antimalarial drugs decrease the effect of:
Antimalarial medications cross the placenta and are considered Category D in pregnancy. Category D implies that they should only be taken during pregnancy if the benefits outweigh the risks. However:
Chloroquine can cause fetal retinopathy and should not be used in pregnancy [4]. Hence, hydroxychloroquine is a safer option for women planning a pregnancy or breastfeeding.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).