Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Autoimmune/autoinflammatory
Original author: Dr Amy Stanway, 2004. Revised and fully updated by Dr Douglas Maslin, Specialist Registrar in Dermatology and Clinical Pharmacology, Addenbrooke’s Hospital, Cambridge, UK. DermNet Editor in Chief: Adjunct A/Prof Amanda Oakley, Dermatologist, New Zealand. Feb 2018; Minor update 2026.
This update was made possible with independent financial support from LEO Pharma A/S.
Introduction
Principles of treatment
General measures
Topical preparation
Ultraviolet treatment
Systemic non-biological medication
Biological therapies
Psoriasis is a common, chronic, immune-mediated skin disease with characteristic red, scaly plaques caused by the excessive proliferation of skin cells.
There are a number of types of psoriasis, including chronic plaque psoriasis, guttate psoriasis, flexural psoriasis, palmoplantar psoriasis, and nail psoriasis.
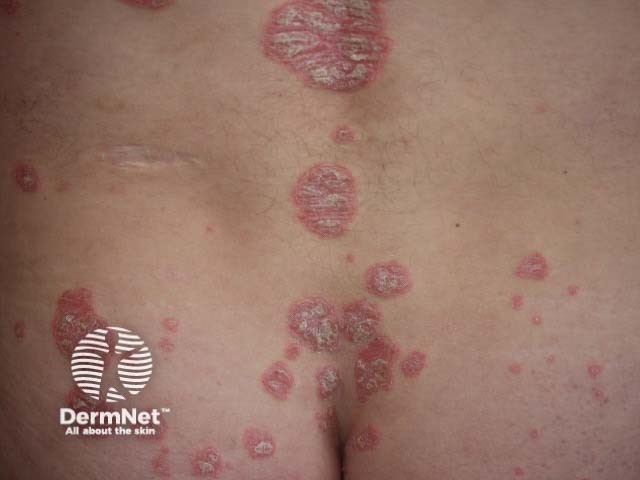
Chronic plaque psoriasis
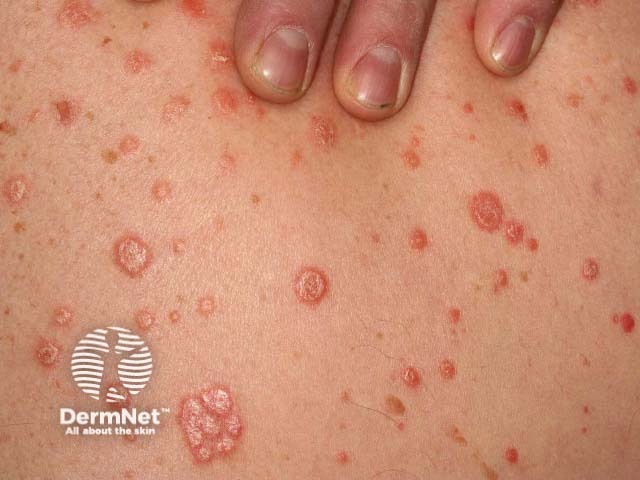
Guttate psoriasis
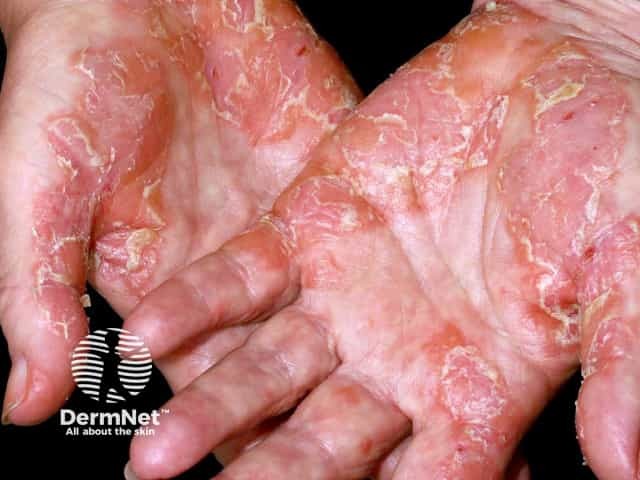
Palmar psoriasis
Despite recent advances in our understanding of the mechanism of how psoriasis develops, psoriasis may be difficult to treat; there is currently no cure and no single treatment works for everyone.
Several treatments may need to be tried before the most suitable regime is established, and different treatments may need to be used concurrently, or in rotation, for best effect or to minimise side-effects.
Treatment of adults with psoriasis includes:
Treatment choice in psoriasis depends on a number of factors. For example:
More details about each form of psoriasis treatment are found on individual topic pages.
Where possible, minimise factors that aggravate or trigger psoriasis, such as stress, streptococcal infections, and certain medications (lithium, beta-blockers, and antimalarial drugs). [see Drug-induced psoriasis]
Health conditions associated with psoriasis include psoriatic arthritis, sleep disturbance, and depression. Treatment for these may help skin disease.
Due to the association between psoriasis and metabolic syndrome, weight loss, smoking cessation, moderation of alcohol intake, and blood pressure control may also lead to improvements in skin disease [1,2].
Sun exposure (heliotherapy) may help to clear psoriasis; in many people, psoriasis improves dramatically during summer months or on sunny holidays.
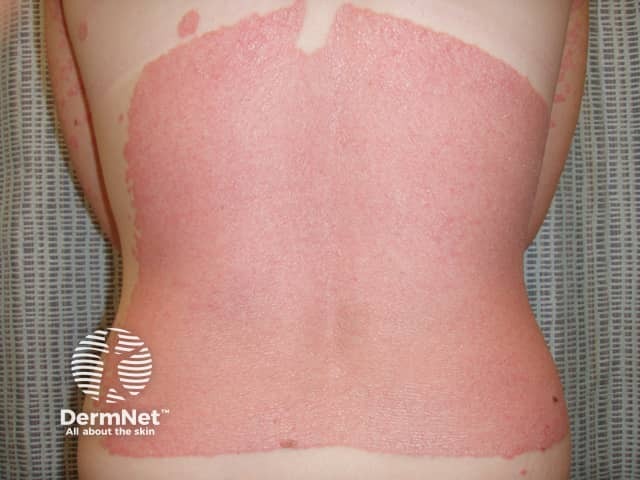
Koebner phenomenon in psoriasis due to sunburn
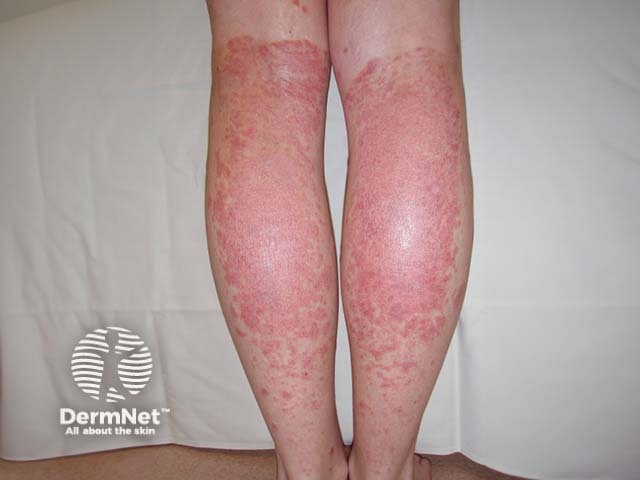
Sunburn and psoriasis
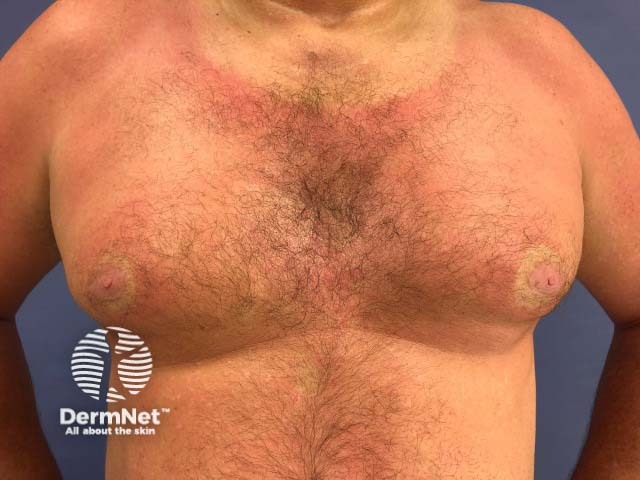
Effect of sun on psoriasis
Soaking in warm water can soften the psoriatic plaques and lift the scale.
One avenue of current research is looking at the skin and gut microbiome (the bacteria living on and in the human body) and whether the alteration of this microbiome may be effective in the treatment of psoriasis [4,5]. To date, probiotics have not been found to help psoriasis.
Relatively small, localised patches of psoriasis may improve with occlusion (eg, using waterproof adhesive dressings).
The regular use of emollients and moisturisers softens psoriasis and adds moisture to the skin. This improves dryness, scaling, and irritation.
Be aware of the flammability of emollients and the risk of slipping in the bath after applying these agents to the feet. Emollients can rarely irritate the skin; this is less likely with ointments than with lotions and creams.
Keratolytic agents can be useful to reduce the thick scale. They may contain urea (5–40%), salicylic acid (0.5–10%) or propylene glycol (for example, propylene glycol 20% in aqueous cream).
Topical steroids are safe and relatively easy to use for plaque psoriasis, scalp psoriasis, flexural psoriasis, sebopsoriasis, and psoriasis affecting the palms and soles. They are not very effective in nail psoriasis.
Topical steroids are available in various strengths and formulations (including steroid impregnated tape). Topical steroids are also used in combination with other agents, such as with:
The selection of a suitable product depends on the site and type of psoriasis.
Potent steroids are often more effective than mild topical steroids, but they have a higher risk of side-effects. They should be used with caution in large areas and for limited periods. They may cause:
Topical steroids are useful for the itch of psoriasis, and work well for the inflammation initially. However with continued use tachyphylaxis develops, and the anti-inflammatory effect wears off.
Topical steroids can be used under medical supervision in pregnancy and, alongside emollients, are generally the first-line treatment of psoriasis in pregnancy.
Intralesional steroid injections may be used for a small number of thickened plaques of psoriasis and in nail psoriasis [6].
Vitamin D-like compounds for psoriasis include calcipotriol, calcitriol, and tacalcitol.
Calcipotriol may be used for chronic plaque psoriasis and scalp psoriasis, whereas calcitriol ointment is often preferred for flexural psoriasis or genital psoriasis (as it is less irritating than calcipotriol).
When combined with ultraviolet (UV) therapy, calcipotriol should be applied after to exposure to UV radiation, because:
Vitamin D-like compounds are best avoided in children under 6 years, and during pregnancy and lactation, due to lack of data regarding their safety.
Calcipotriol is available in combination with a very potent topical steroid, betamethasone dipropionate as a gel/ointment or foam.
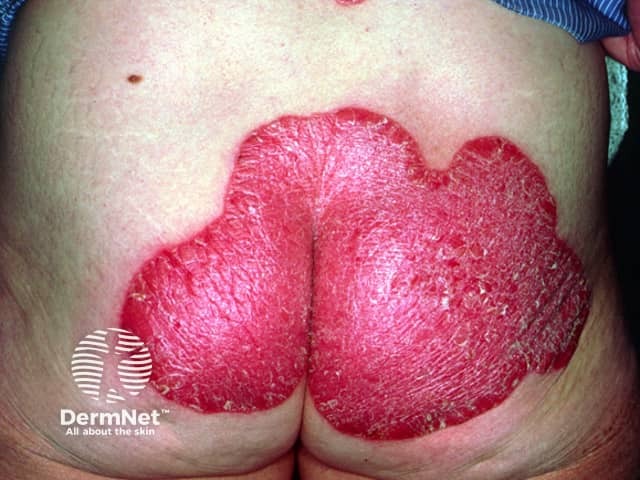
Pre-treatment
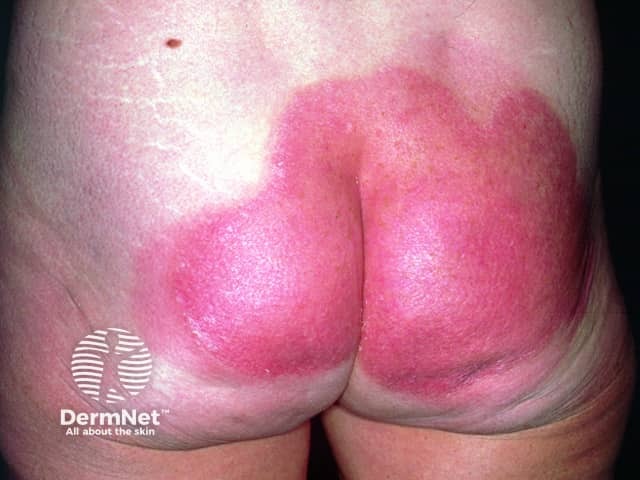
Post-treatment
Coal tar may be applied as solutions, lotions, creams, ointments, gels, and shampoos.
Care needs to be taken following the application of coal tar treatments.
Dithranol (also called anthralian or anthralin) is occasionally recommended as a treatment for chronic plaque psoriasis. It can be very effective but dithranol treatment has a number of practical drawbacks and so is less frequently prescribed.
The method of application is complex; it is usually given as ‘short contact' therapy.
The calcineurin inhibitors are tacrolimus ointment and pimecrolimus cream.
Tazarotene is a topical retinoid, which may be applied once daily to plaque psoriasis as a 0.05% or 0.1% cream. [8].
The most common side-effect is skin pain and local irritation. It is not currently available in New Zealand (July 2018).
Topical roflumilast 0.3% cream (Zorvye™) was approved by the FDA in 2022 for the treatment of plaque psoriasis in patients aged 12 years and older. This is the first topical PDE4 inhibitor approved for use in psoriasis.
Tapinarof 1% cream (VTAMA®) is a once-daily, novel, topical agent for patients with mild, moderate, and severe plaque psoriasis, first approved by the US Food and Drug Administration (FDA) in May 2022.
Off-label use of the phosphodiesterase-4 topical inhibitor crisaborole has been shown to be effective for flexural and facial psoriasis.
Phototherapy is the use of UV radiation to treat skin disorders, and this can be very effective in the treatment of psoriasis. It is generally reserved for cases where topical therapy has been ineffective or too much of the skin surface is involved to treat psoriasis effectively with topical agents. It is administered in cabinets at specialised centres, and a treatment course for psoriasis will usually consist of 2–3 treatments per week for 20–30 treatments.
The need for regular travel to a phototherapy centre can make this option difficult for some patients. The beneficial effects may be short-lived.
Narrowband ultraviolet B (UVB) (311–312 nm wavelengths) is also known as TL01 light therapy (after the type of fluorescent tubes used).
Psoralens plus long wave ultraviolet A (UVA) radiation, (known as photochemotherapy), can be applied to the whole body by giving an oral psoralen in tablet form 2 hours prior to treatment.
Systemic (oral or injectable) medication may be required to treat psoriasis when:
The selection of the appropriate medication is specific to each individual patient, as each agent carries its own risks and benefits.
As a rule, systemic steroids should be avoided as they can make psoriasis worse or unstable and difficult to control with other treatments.
Methotrexate is an immunomodulatory and anti-inflammatory treatment used to treat psoriasis, psoriatic arthritis, and Crohn disease.
The dose of methotrexate is often adjusted over the first few weeks or months of treatment. There are various regimens.
Side-effects of methotrexate include nausea, tiredness, mouth ulcers, and diarrhoea.
Ciclosporin is an immunosuppressant used short-term to treat atopic eczema and psoriasis.
Ciclosporin has a rapid onset of action, making it useful in severe plaque psoriasis or pustular psoriasis.
Important side-effects of ciclosporin include hypertension, renal failure, susceptibility to infection, and increased risk of skin cancer.
Acitretin is a vitamin A-like compound or retinoid that is particularly effective for palmoplantar psoriasis.
Dose-related side-effects of acitretin include dry lips, peeling palms and soles, thinning hair, tiredness, and muscle pains.
Apremilast is a phosphodiesterase-4 inhibitor used to treat psoriasis and psoriatic arthritis. It is not funded in New Zealand (July 2018).
Side effects of apremilast include nausea and diarrhoea. There have also been reports of suicidal behaviour.
Fumaric acid esters are immunosuppressants. They are not available or funded in New Zealand.
Side-effects include nausea, diarrhoea, stomach cramps, flushing, and headaches. A rare but serious side-effect is a nervous system viral infection (progressive multifocal leukoencephalopathy).
Fumaric acid esters are not to be used during pregnancy due to fetal harm.
Other non-biological oral medications for psoriasis include:
Biological therapies or biologics are monoclonal antibodies or recombinant proteins targeted at specific components of the immune system. They are often very effective treatments for psoriasis.
Each available biologic has individual risks and benefits. Novel biological therapies are under development. Currently available biologics and those under development include:
The biologicals for psoriasis are given by subcutaneous injection, with the exception of infliximab, which is administered intravenously. Infliximab is a chimera of mouse and human protein and can result in infusion reactions and antibody formation.
Biosimilars are drugs that are nearly identical to an original biological medication that has come off patent [15] and are available at a reduced cost. Biosimilars are available for infliximab and etanercept and others are under development (July 2018).
Oral drugs under development include small molecule compounds targeting signalling pathways, such as JAK inhibitors and PDE inhibitors. As they are low molecular weight, topical formulations are being investigated.
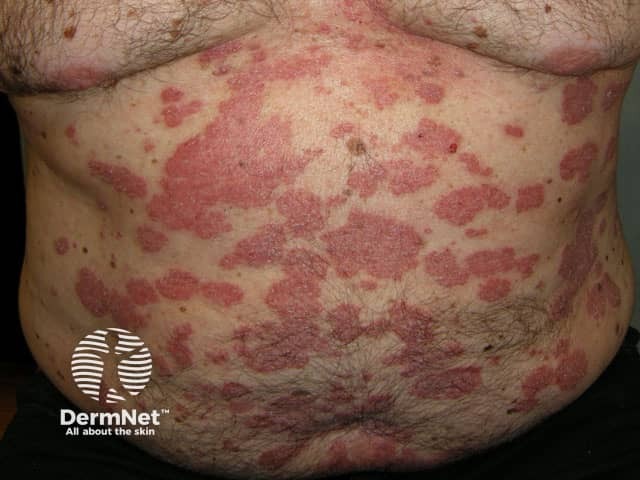
Before treatment
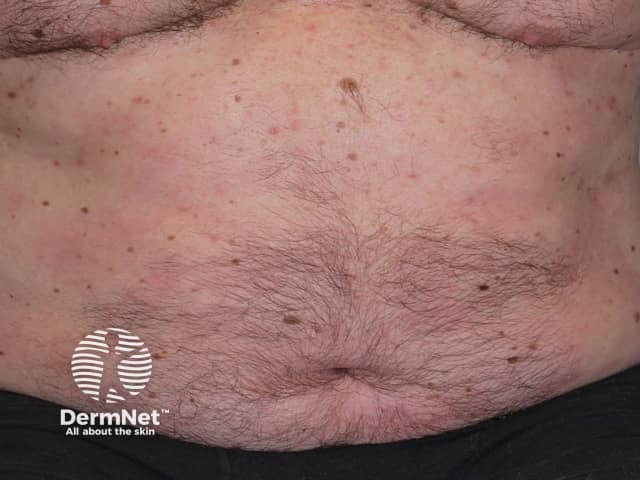
After treatment